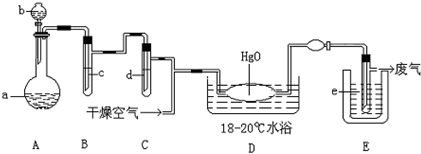
��Ŀ����
 ��2009?�ϻ���ģ�⣩��100mLBaCl2��AlCl3��FeCl3�Ļ����ҺA�У���μ���Na2SO4��NaOH�Ļ����ҺB�������ij����ͼ�����ҺB�������ϵ��ͼ��ʾ��
��2009?�ϻ���ģ�⣩��100mLBaCl2��AlCl3��FeCl3�Ļ����ҺA�У���μ���Na2SO4��NaOH�Ļ����ҺB�������ij����ͼ�����ҺB�������ϵ��ͼ��ʾ����1��������B��Һ110mLʱ����Һ�еij�����
BaSO4��Fe��OH��3
BaSO4��Fe��OH��3
���ѧʽ������2����ҺB��Na2SO4��NaOH�����ʵ���Ũ��֮��
1��1
1��1
����90mL��100mL֮�����10mL B��Һʱ�����������ӷ�Ӧ����ʽ��Ba2++SO42-=BaSO4����Al��OH��3+OH-=AlO2-+2H2O
Ba2++SO42-=BaSO4����Al��OH��3+OH-=AlO2-+2H2O
����3����A��B��Һ�и����ʵ����ʵ���Ũ�������±���
| ���� | Na2SO4 | NaOH | BaCl2 | AlCl3 | FeCl3 |
| Ũ��mol?L-1 | 10 10 |
10 10 |
10 10 |
2 2 |
1 1 |
������ͼ�е�һ���������ɵij���ΪBaSO4��Fe��OH��3��Al��OH��3���Ҽ���B��Һ90mLʱ��Fe3+��Al3+ǡ�ó�����ȫ��
ͼ�еڶ�������Ϊƽ��ֱ�ߣ����������ʵ���û�����������������Al��OH��3���ܽ��BaSO4�ļ����������������ܽ��Al��OH��3�����ʵ��������ɵ�BaSO4�����ʵ�����ȣ�ͼ�е����������У�����B��Һ100mLʱ��Ba2+ǡ�ó�����ȫ�������Al��OH��3�����ܽ⣬BΪ110mLʱ��������Al��OH��3����ȫ�ܽ⣮
ͼ�е��Ķ������У�����ΪFe��OH��3��BaSO4��1.1mol��
ͼ�еڶ�������Ϊƽ��ֱ�ߣ����������ʵ���û�����������������Al��OH��3���ܽ��BaSO4�ļ����������������ܽ��Al��OH��3�����ʵ��������ɵ�BaSO4�����ʵ�����ȣ�ͼ�е����������У�����B��Һ100mLʱ��Ba2+ǡ�ó�����ȫ�������Al��OH��3�����ܽ⣬BΪ110mLʱ��������Al��OH��3����ȫ�ܽ⣮
ͼ�е��Ķ������У�����ΪFe��OH��3��BaSO4��1.1mol��
����⣺��Һ��ֻ��Fe3+����ȫ�����Ժ�Al3+�ſ�ʼ������
ͼ�е�һ���������ɵij���ΪBaSO4��Fe��OH��3��Al��OH��3���Ҽ���B��Һ90mLʱ��Fe3+��Al3+ǡ�ó�����ȫ��
��η�Ӧ�����ӷ���ʽΪBa2++SO42-=BaSO4����Fe3++3OH-=Fe��OH��3����Al3++3OH-=Al��OH��3����
ͼ�еڶ�������Ϊƽ��ֱ�ߣ����������ʵ���û�����������������Al��OH��3���ܽ��BaSO4�ļ����������������ܽ��Al��OH��3�����ʵ��������ɵ�BaSO4�����ʵ�����ȣ��ʼ�������ʣ�n��NaOH��=n��Na2SO4������η�Ӧ�����ӷ���ʽΪ��Al��OH��3+OH-=AlO2-+2H2O��Ba2++SO42-=BaSO4����
ͼ�е����������У�����B��Һ100mLʱ��Ba2+ǡ�ó�����ȫ�������Al��OH��3�����ܽ⣬BΪ110mLʱ��������Al��OH��3����ȫ�ܽ⣬�ܽ���0.1molAl��OH��3��������10mLB��Һ����c��NaOH��=10mol/L������Na2SO4Ϊ0.1mol����c��Na2SO4��=10mol/L����������Ӧ���̹�����BaSO4Ϊ0.1��10=1��mol������A��Һ��c��Ba2+��=10mol/L��Al��OH��3�ܽ�����ӷ���ʽΪ��Al��OH��3+OH-=AlO2-+2H2O��
ͼ�е��Ķ������У�����ΪFe��OH��3��BaSO4��1.1mol����Fe��OH��3Ϊ0.1mol��A��c��Fe3+��=1mol/L���ڶ����������߹�����B��Һ20mL�����ܽ��Al��OH��3Ϊ0.2mol����A��c��Al3+��=2mol?L-1��
�ʴ�Ϊ��
��1��BaSO4��Fe��OH��3��
��2��1��1��Ba2++SO42-=BaSO4����Al��OH��3+OH-=AlO2-+2H2O��
��3��
ͼ�е�һ���������ɵij���ΪBaSO4��Fe��OH��3��Al��OH��3���Ҽ���B��Һ90mLʱ��Fe3+��Al3+ǡ�ó�����ȫ��
��η�Ӧ�����ӷ���ʽΪBa2++SO42-=BaSO4����Fe3++3OH-=Fe��OH��3����Al3++3OH-=Al��OH��3����
ͼ�еڶ�������Ϊƽ��ֱ�ߣ����������ʵ���û�����������������Al��OH��3���ܽ��BaSO4�ļ����������������ܽ��Al��OH��3�����ʵ��������ɵ�BaSO4�����ʵ�����ȣ��ʼ�������ʣ�n��NaOH��=n��Na2SO4������η�Ӧ�����ӷ���ʽΪ��Al��OH��3+OH-=AlO2-+2H2O��Ba2++SO42-=BaSO4����
ͼ�е����������У�����B��Һ100mLʱ��Ba2+ǡ�ó�����ȫ�������Al��OH��3�����ܽ⣬BΪ110mLʱ��������Al��OH��3����ȫ�ܽ⣬�ܽ���0.1molAl��OH��3��������10mLB��Һ����c��NaOH��=10mol/L������Na2SO4Ϊ0.1mol����c��Na2SO4��=10mol/L����������Ӧ���̹�����BaSO4Ϊ0.1��10=1��mol������A��Һ��c��Ba2+��=10mol/L��Al��OH��3�ܽ�����ӷ���ʽΪ��Al��OH��3+OH-=AlO2-+2H2O��
ͼ�е��Ķ������У�����ΪFe��OH��3��BaSO4��1.1mol����Fe��OH��3Ϊ0.1mol��A��c��Fe3+��=1mol/L���ڶ����������߹�����B��Һ20mL�����ܽ��Al��OH��3Ϊ0.2mol����A��c��Al3+��=2mol?L-1��
�ʴ�Ϊ��
��1��BaSO4��Fe��OH��3��
��2��1��1��Ba2++SO42-=BaSO4����Al��OH��3+OH-=AlO2-+2H2O��
��3��
| ���� | Na2SO4 | NaOH | BaCl2 | AlCl3 | FeCl3 |
| Ũ��mol?L-1 | 10 | 10 | 10 | 2 | 1 |
���������⿼�����ӷ���ʽ���йؼ��㣬��Ŀ�ѶȽϴ�ע�����ͼ����ȷ�жϸ��ε����ӷ�Ӧ����ϸ��εĸ������ʵ������м��㣮

��ϰ��ϵ�д�
�����Ŀ