��Ŀ����
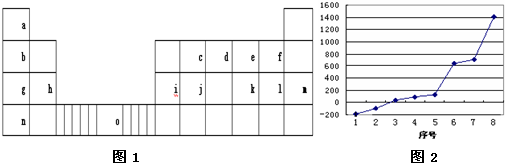
15����ͼ1��Ԫ�����ڱ���һ���֣�ͼ�����е���ĸ�ֱ����һ�ֻ�ѧԪ�أ��Իش��������⣺
��1����д��Ԫ��O�Ļ�̬ԭ�ӵ����Ų�ʽ1s22s22p63s23p63d64s2����
��2����������8��Ԫ�ذ������۵�ߵ͵�˳����ͼ2��������š�8������Si����Ԫ�ط��ţ������е縺��������l ����ͼ2����ţ���һ�����ܽ���������֮��ĵ�������Ԫ����3�֣�
��3����jԭ�Ӹ�cԭ����1��1������϶��γɵľ��壬�����뾧��j��ͬ����������۵���ߵ���SiC���ѧʽ�����Դӽṹ�Ƕȼ��Խ��ͣ���SiC�����뾧��Si����ԭ�Ӿ��壬����C��ԭ�Ӱ뾶С��SiC��C-Si�������Ⱦ���Si��Si-Si�����̣����ܴ�����۷е�ߣ�
��4��k l2�ĵ���ʽ��
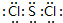 �����ڳ����³�Һ̬���γɾ���ʱ�����ڷ��Ӿ��壮
�����ڳ����³�Һ̬���γɾ���ʱ�����ڷ��Ӿ��壮
���� ��Ԫ���������ڱ��е�λ�ÿ�֪��aΪH��bΪLi��cΪC��dΪN��eΪO��fΪF��gΪNa��hΪMg��iΪAl��jΪSi��kΪS��lΪCl��mΪAr��nΪK��oΪFe��
��1��oΪFe��ԭ�Ӻ��������Ϊ26�������������ԭ����д��������Ų���
��2����������8��Ԫ�صĵ����γɽ������塢ԭ�Ӿ��塢���Ӿ��壬ԭ�Ӿ���Si���۵����
ͬ������ԭ���������Ԫ�ص縺������ϡ��������⣩��
ͬ������ԭ����������Ԫ�ص�һ�����ܳ��������ƣ�����A����A��Ԫ�ص�һ�����ܸ���ͬ��������Ԫ�أ�
��3��SiC�뾧��Si��Ϊԭ�Ӿ��壬����Խ�̣�����Խ���۵�Խ�ߣ�
��4��SCl2������Sԭ����Clԭ��֮���γ�1�Թ��õ��Ӷԣ����ڳ����³�Һ̬���γɾ���ʱ�����ڷ��Ӿ��壮
��� �⣺��Ԫ���������ڱ��е�λ�ÿ�֪��aΪH��bΪLi��cΪC��dΪN��eΪO��fΪF��gΪNa��hΪMg��iΪAl��jΪSi��kΪS��lΪCl��mΪAr��nΪK��oΪFe��
��1��oΪFe��ԭ�Ӻ��������Ϊ26����������Ų�Ϊ��1s22s22p63s23p63d64s2����
�ʴ�Ϊ��1s22s22p63s23p63d64s2��
��2����������8��Ԫ�صĵ����γɽ������塢ԭ�Ӿ��塢���Ӿ��壬ԭ�Ӿ���Si���۵����
ͬ������ԭ���������Ԫ�ص縺������ϡ��������⣩����ClԪ�ص縺�����Ϊ����l��
ͬ������ԭ����������Ԫ�ص�һ�����ܳ��������ƣ�����A����A��Ԫ�ص�һ�����ܸ���ͬ��������Ԫ�أ��ʵ�һ�����ܽ���������֮��ĵ�������Ԫ���У�Mg��Si��S��
�ʴ�Ϊ��Si��l��3��
��3��SiC�;���Si������ԭ�Ӿ��壬ԭ�Ӿ�����۷е�ȡ���ڹ��ۼ���ǿ�ȣ�����C��ԭ�Ӱ뾶С��SiC��C-Si�������Ⱦ���Si��Si-Si�����̣����ܴ�����۷е�ߣ�
�ʴ�Ϊ��SiC�� ��SiC�����뾧��Si����ԭ�Ӿ��壬����C��ԭ�Ӱ뾶С��SiC��C-Si�������Ⱦ���Si��Si-Si�����̣����ܴ�����۷е�ߣ�
��4��SCl2������Sԭ����Clԭ��֮���γ�1�Թ��õ��Ӷԣ�����ʽΪ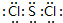 �����ڳ����³�Һ̬���γɾ���ʱ�����ڷ��Ӿ��壬
�����ڳ����³�Һ̬���γɾ���ʱ�����ڷ��Ӿ��壬
�ʴ�Ϊ��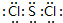 �����ӣ�
�����ӣ�
���� ����ʽ�����ʽṹ�Ŀ��飬�漰Ԫ�����ڱ����縺�ԡ������ܡ��������������ʡ���������Ų�������ʽ�ȣ�ע������ͬ���ڵ�һ�������쳣������Ѷ��еȣ�

| A�� | 25��ʱ������Mg��OH��2��20ml0.01 mol•L-1��ˮ�е�KSP��20mL0.01 mol•L-1NH4Cl��Һ�е�KSPС | |
| B�� | 25��ʱ������Mg��OH��2��Һ�뱥��MgF2��Һ��ȣ�ǰ�ߵ�C��Mg��2+�� | |
| C�� | 25��ʱ����Mg��OH��2������Һ����NaF��Һ��Mg��OH��2������ת����ΪMgF2 | |
| D�� | 25��ʱ����Mg��OH��2������Һ����������NH4Cl���壬C��Mg2+������ |
| A�� | Cl2 | B�� | NO2 | C�� | SO2 | D�� | NH3 |
| A�� | 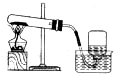 ��ȡ���ռ�O2 ��ȡ���ռ�O2 | B�� | 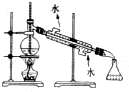 ����ˮ���� ����ˮ���� | ||
| C�� | 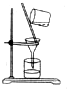 ��ȥNaCl�е���ɳ ��ȥNaCl�е���ɳ | D�� | 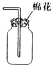 �ռ�NO �ռ�NO |
| A�� | ����ͨ������������Һ�� 2Cl2+2OH-�T3Cl-+ClO-+H2O | |
| B�� | ������ʯ��ʯ��Ӧ��2H++CO32-�TH2O+CO2�� | |
| C�� | п��ϡ���ᷴӦ��Zn+2H+�TZn2++H2�� | |
| D�� | ����ͭ��Һ������������Һ��Ӧ��Ba2++SO42-�TBaSO4�� |
| A�� | 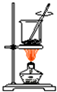 �����ճɻ� �����ճɻ� | |
| B�� | 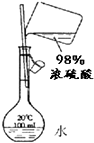 ����ϡ���� ����ϡ���� | |
| C�� |  ����Ⲣ���ձ� ����Ⲣ���ձ� | |
| D�� |  XΪCCl4������������HCl������ֹ���� XΪCCl4������������HCl������ֹ���� |
| A�� | 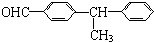 | B�� | 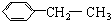 | ||
| C�� | CH2=CHCH=CHCH=CH2 | D�� | ��CH3��2CH-C��C-CH=CHCH3 |
| A�� | ���ʯΪ��״�ṹ���ɹ��ۼ��γɵ�̼ԭ�ӻ��У���С�Ļ�����6��̼ԭ�� | |
| B�� | �Ȼ��ƾ����У�ÿ��Na+��Χ������ȵ�Na+����12�� | |
| C�� | �ɱ������У�ÿ��CO2������Χ����12��CO2���� | |
| D�� | ����ͭ�����������ܶѻ��ṹ������þ���������������ܶѻ��ṹ |