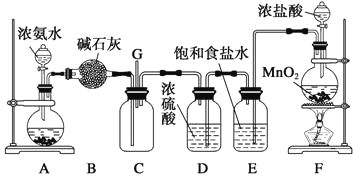
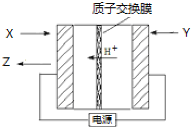
��Ŀ����
����Ŀ��ij�о���ѧϰС�齫����װ����ͼ���ӣ�D��F��X��Y ���Dz��缫��C��E�����缫����Դ��ͨ�������е����̪��Һ����F�������Ժ�ɫ������˵������ȷ���� �� ��
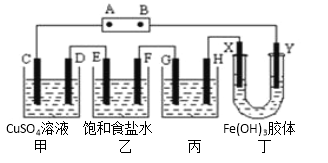
A����ԴB ���������Ǹ���
B����װ���е�ⷴӦ���ܻ�ѧ����ʽ��: CuSO4+Fe![]() Cu+ FeSO4
Cu+ FeSO4
C����������Һ��������ͬһʱ��C��D�缫�ϲμӷ�Ӧ�ĵ��ʻ����ɵĵ��ʵ����ʵ���֮����1:1
D����׳�����Һ������ڵ��ǰ����500ml�����ҳ���������������Ϊ4.48L����״����ʱ���׳������������ʵ����ʵ���Ũ��Ϊ0.2mol/L
���𰸡�D
��������
���������A��F���Ժ�ɫ��˵������OH����OH�����������������ݵ��ԭ����BΪ������AΪ��������˵����ȷ��B��C�ӵ�Դ��������C��������Fe��2e��=Fe2����DΪ��������ӦʽΪCu2����2e��=Cu���ܵ缫��ӦʽΪ��CuSO4+Fe![]() Cu+ FeSO4����˵����ȷ��C��C����ӦʽΪFe��2e��=Fe2����D����ӦʽΪCu2����2e��=Cu��ͨ��������ȣ�������ĺͲ������ʵ����ʵ���֮��Ϊ1��1����˵����ȷ��D���ҳ�E�缫��ӦʽFe��2e��=Fe2����F�缫��Ӧʽ2H2O��2e��=H2����2OH�����ҳ��в����������Ϊ4.48L��ת�Ƶ��ӵ����ʵ���Ϊ0.4mol��ͨ��������ȣ���˲���FeSO4�����ʵ���Ϊ0.2mol�����Ũ��Ϊ0.2/500��10��3mol��L��1=0.4mol��L��1����˵������
Cu+ FeSO4����˵����ȷ��C��C����ӦʽΪFe��2e��=Fe2����D����ӦʽΪCu2����2e��=Cu��ͨ��������ȣ�������ĺͲ������ʵ����ʵ���֮��Ϊ1��1����˵����ȷ��D���ҳ�E�缫��ӦʽFe��2e��=Fe2����F�缫��Ӧʽ2H2O��2e��=H2����2OH�����ҳ��в����������Ϊ4.48L��ת�Ƶ��ӵ����ʵ���Ϊ0.4mol��ͨ��������ȣ���˲���FeSO4�����ʵ���Ϊ0.2mol�����Ũ��Ϊ0.2/500��10��3mol��L��1=0.4mol��L��1����˵������

����Ŀ����֪�±���������ij��ͬ���칹���һ�ȴ��ﶼֻ��һ�ֽṹ�����ݱ������ʵķ���ʽ�Ʋ���е�����Ӧ��Ϊ�� ��
1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
CH4 | C2H6 | C5H12 | C8H18 | C17H36 | ���� | C53H108 | C80H162 |
A. C22H46 B. C26H54 C. C30H62 D. C32H66