��Ŀ����
ʳ�����ճ�����ı���Ʒ��Ҳ����Ҫ�Ļ���ԭ�ϡ�
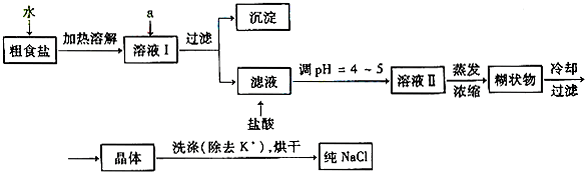
(1)��ʳ�γ���������Ca2����Mg2����SO42�����������ӣ�ʵ�����ᴿNaCl���������£�
�ṩ���Լ�������Na2CO3��Һ ����K2CO3��Һ NaOH��Һ BaCl2��Һ Ba(NO3)2��Һ
(1)������ȥ��Һ���е�Ca2����Mg2����SO42����ѡ��A���������Լ������μ�˳������Ϊ (ֻ�ѧʽ)��������Һ��SO42��������ȫ�IJ���Ϊ ��
�ڼ������Ŀ����(�����ӷ���ʽ��ʾ) ��
�����������У��ס����������̾�Ҫ�õ��������������������÷ֱ��Ǽ� ���� ��
(2)��ҵ���ö��Ե缫��ⱥ��ʳ��ˮ����ȡ���ᣬд����ⱥ��ʳ��ˮ�Ļ�ѧ����ʽ ������ֽ������ʳ��ˮ��������ķ����� ��
��1����BaCl2��NaOH ��Na2CO3��NaOH��BaCl2��Na2CO3
�Դ�Ƭ�̣�������������ϣ��ϲ������Һ�����ϲ���Һ�м����μ��Ȼ�����Һ���������ɣ��������ȫ��
��OH����H��=H2O CO32����2H��=H2O��CO2��
�۽�������
��2��2NaCl��2H2O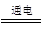 2NaOH��H2����Cl2��
2NaOH��H2����Cl2��
��ʪ�����ɫʯ����ֽ���������������У���ֽ�ȱ�죬����ɫ��֤������������(����ʪ���KI-������ֽ���������������У���ֽ����ɫ)
���������������1������Ca2+��Na2CO3��Һ������Mg2+��NaOH��Һ������SO42-��BaCl2��Һ����Ҫע������Լ���������ҹ������Լ��������������ȥ�������Լ��μӵ�˳����BaCl2��NaOH ��Na2CO3��NaOH�� BaCl2��Na2CO3������SO42-������ȫ�IJ�������Ϊ����Һ���ã����ϲ���Һ�м����μ��Ȼ�����Һ���������ɣ�˵��SO42-��ȫ��������Һ�м�����������dz�ȥ������CO32-��OH-���ܽ�ʱ�ò��������裬�ӿ��ܽ��ٶȡ�����ʱ�ò�����������
��2����ⱥ��ʳ��ˮ�����������ơ�������������������������������ʪ�����-KI��ֽ���顣
���㣺��ѧʵ�� ��� Ԫ�ؼ��仯����
����������ʱҪע���Լ������˳���������Լ���������������Լ��������������ȥ��

 ��У����ϵ�д�
��У����ϵ�д�