��Ŀ����
��08ɽ���߿���̾�����14�֣�A�DJ����ѧ��ѧ�г����ļ������ʣ�����֮���ת����ϵ��ͼ��ʾ����֪������AΪ���嵥�ʣ�BΪ����ɫ��ĩ��C��F��IΪ��̬���ʣ�E�ڳ�����ΪҺ�壬��E����C��F�ϳɣ�J��������ǰ�����в�����ɱ����������
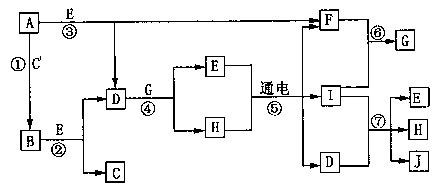 |
��1����EΪ_________��
��2��д����Ӧ�ߵ����ӷ���ʽ__________________________��
��3����AlCl3��Һ�м�����������B��д����Ӧ�Ļ�ѧ����ʽ_____________________��
��4����PtΪ�缫���μ���������̪��H������Һ������_____________���������������������Һ����ɫ��Ϊ��ɫ����ԭ����_________________��
������1��H2O ��2�֣�
��2��Cl2+2OH��=Cl��+ClO��+H2O ��3�֣�
��3��4AlCl3+6Na2O2+6H2O=4Al(OH)3��+12NaCl+3O2����3�֣�
(4)���� ��3�֣� ����������H+�õ����Ӳ���H2���ƻ���ˮ�ĵ���ƽ�⣬�ٽ�ˮ�������룬������Һ��c(OH��)>c(H+)����Һ�ʼ��ԣ���������������Һ��Ϊ��ɫ��3�֣�
���������������е��ƶ��⣬������ѧ�����ƶ����������⣬������µĿ��Դ���е�Ҫ�������ˣ���˵����ʿ����˵��֪ʶ�������ƶ���Ľ���ؼ�����ͻ�ƿڣ������ͻ�ƿھ�����Ŀ�и����ĸ�Ԫ�ؼ��䵥�ʡ�����������ʡ�

 ����ѵ�����⿼ϵ�д�
����ѵ�����⿼ϵ�д���08ɽ���߿���̾�����8�֣�2007��10��������ά�Ƽ����140������1867���ڰ������粩�����ϣ��������ʽ����Ϊ����ά�Ƽ���÷��㷺Ϊ����������ã����й��ĺ����Ƽ��ȡ��������ά�������������ǣ�
 |
����ά����ʵ��������������ʳ��������ֻ��70���Ҹ���ƷCaCl2û����;����Ⱦ������
1940���ҹ���ѧ�Һ�°������������ʵ�飬�����ˡ�����ά�����ļ��������������ԸĽ���ʳ�������ʴ�96���õ��˴�����Ȼ�����ֲ�Ʒ������Ϊ�������Ƽ���������ԭ���ǣ�
����30�桫50��ı���ʳ��ˮ�У���ͨ�백���ͣ���ͨ��CO2��̼�����Ƴ�����
�ڹ��ˣ����������ȶ��ø���Ʒ��
������Һ�м���ϸʳ��ĩ����10�桫15�棬ʹNH4Cl���������ˣ���ҺΪ����ʳ��ˮ��
������������⣺
��1���ڰ��ı���NaCl��Һ��ͨ��CO2��������Ӧ����ʽ��
______ ___________��
��2��������NaCl��Һ��ͨ��CO2��NaHCO3��ԭ����____________________��
��3������С�մ��ĸҺ�м�����ʯ�ҵķ�Ӧ����ʽ��_______________��
��4���ڡ������Ƽ���У�_______���ʿ���ѭ�����ã��ڡ�����ά������___________���ʿ���ѭ�����á�
��5���������Ƽ���롰����ά����ȣ����ŵ���___________��