��Ŀ����
Ϊ��֤���Ҵ������к�����ԭ�ӣ�ijС���������ͼװ�ý���ʵ���о���
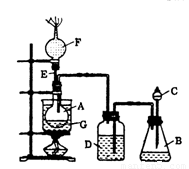
ͼ���ձ�A����ˮԡ������ƿB��ʢ���Լ�X����Һ©��C���ƿD�о�ʢ��Ũ���ᣬE��һ��ֱ���ܣ������F���������Ƶļ�ʯ�ң���ƿG��װ����ˮ�Ҵ���һЩ���ư�ɫ��ˮ�η�ĩY����C����������Ũ������������B�����Լ�X���ã�֮����Dƿ�ĵ��ܿڲ����������ݣ�ˮԡ����Gƿ�������ֹ۲쵽Gƿ���Լ�Y��ɫ��F���ų������徭�������ܵ�ȼ����ش��������⣺
��1��Bƿ����װ���Լ�X�����µ�_________����ѡ����ĸ����
A������ʳ��ˮ B��MnO2��NaCl����� C��Ũ����
��2��C��Ũ�������Ҫ������____________________________��B���ݳ�������ɷ���
__________________��
��3��D����װŨ�����������__________________��
��4��Gƿ�з����л���Ӧ�Ļ�ѧ����ʽ��___________________________��
��5��F���������Ƶļ�ʯ�ҵ�Ŀ����__________________________��E���ܳ����е����Ļ��������⣬�ڱ�ʵ���л����е�������___________��
��6����ˮ�η�ĩY�Ļ�ѧʽ��_______________����ʵ����֤���Ҵ������к�����ԭ�ӵ�������_______________________________��
�����24�֣�
��1��C ��2�֣���
��2���ܽ���ȣ��ٽ�HCl�ӷ� ��1�֣��� HCl��ˮ���� ��2�֣���
��3������ ��1�֣���
��4��CH3CH2OH��HCl CH3CH2Cl��H2O ��2�֣���
CH3CH2Cl��H2O ��2�֣���
��5����ֹ�����е�ˮ�������� ��1�֣��� �������� ��1�֣���
��6��CuSO4 ��1�֣��� �Լ�Y�ɰ�ɫ����ɫ ��1�֣���
����������