��Ŀ����
 ʵ�����Ʊ������飨C2H5Br����װ�úͲ�����ͼ������֪������ķе�38��4�棩
ʵ�����Ʊ������飨C2H5Br����װ�úͲ�����ͼ������֪������ķе�38��4�棩
�ټ��װ�õ������ԣ���װ��ͼ��ʾ��U�ιܺʹ�
�ձ��м����ˮ��
����Բ����ƿ�м���10mL95%�Ҵ���28mL80%Ũ
���ᣬȻ�������ϸ��13g�廯�ƺͼ������Ƭ��
��С�ļ��ȣ�ʹ���ַ�Ӧ��
�ش��������⣺
��1����ʵ����ȡ������Ļ�ѧ����ʽΪ��
____________________________����������������
��2����Ӧʱ���¶ȹ��ߣ��ɿ����к���ɫ������������������ʽΪ_______ ____��ͬʱ��������һ�ֵ���ɫ���塣
��3��U���ڿɹ۲쵽��������___________________________��
��4����Ӧ������U�ι��д��Ƶ�C2H5Br���ػ�ɫ��Ϊ�˳�ȥ�ֲ�Ʒ�е����ʣ���ѡ�������Լ��е�_______________������ĸ��
A��NaOH��Һ B��H2O
C��Na2SO3��Һ D��CCl4
�������Ҫ����������______________�����������ƣ���Ҫ��һ���Ƶô�����C2H5Br������ˮϴ��Ȼ�������ˮCaCl2���ٽ���_______________����������ƣ���
��5�����м���ʵ�鲽�裬�����ڼ�������������Ԫ�أ�����ȷ�IJ���˳���ǣ�ȡ���������飬Ȼ��__________________������ţ���
�ټ��ȣ��ڼ���AgNO3��Һ���ۼ���ϡHNO3�ữ���ܼ���NaOH��Һ������ȴ
��6�������飨C2H5Br����һ��̼��ͬϵ�����������ƵĴ���Һ���ܷ�����Ӧ��д���÷�Ӧ�Ļ�ѧ����ʽ����������������������������������������������������������
��16�֣�ÿ��2�֣�
��1��C2H5OH+NaBr+H2SO4![]() NaHSO4+C2H5Br+H2O
NaHSO4+C2H5Br+H2O
��2��Br2 ��3������״Һ�����ɡ�
��4��c�� ��Һ©���� ���� ��5���ܢ٢ݢۢ�
 |
��6��CH3CH2CH2Br��NaOH CH3��CH��CH2����NaBr��H2O

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�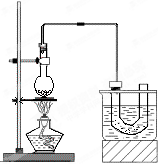 ��ʵ�����Ʊ������飨C2H5Br����װ�úͲ�����ͼ������֪������ķе�38.4�棩
��ʵ�����Ʊ������飨C2H5Br����װ�úͲ�����ͼ������֪������ķе�38.4�棩 ��֪��CH3CH2OH+NaBr+H2SO4��Ũ��
��֪��CH3CH2OH+NaBr+H2SO4��Ũ�� ������ķе���38.4�棬�ܶ���1.46g/cm3����ͼΪʵ�����Ʊ��������װ��ʾ��ͼ���г���������ȥ��������G��ʢ����ˮ��ʵ��ʱѡ�õ�ҩƷ�У��廯�ơ�95%�Ҵ���ŨH2SO4���Ʊ������б߷�Ӧ��������������������ˮ���ռ�����ã�
������ķе���38.4�棬�ܶ���1.46g/cm3����ͼΪʵ�����Ʊ��������װ��ʾ��ͼ���г���������ȥ��������G��ʢ����ˮ��ʵ��ʱѡ�õ�ҩƷ�У��廯�ơ�95%�Ҵ���ŨH2SO4���Ʊ������б߷�Ӧ��������������������ˮ���ռ�����ã�