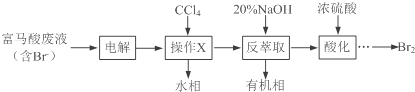
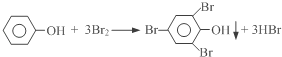
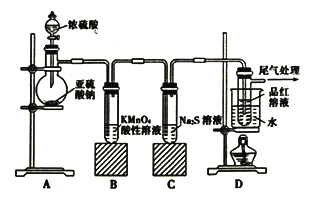
��Ŀ����
����Ŀ����������Ҫ�Ĺ�ҵԭ�ϡ�
(1)��״���£���500L��������ˮ�γ�1L��ˮ����˰�ˮ�����ʵ���Ũ��Ϊ______mol/L(������λ��Ч���֣����ð�ˮ��������ն�����������Ϊ_________��
(2)���Ĵ���������Ҫ�Ĺ�ҵ�������˷�Ӧ�����Ϳ����Ļ����ͨ�����ȵIJ���Ͻ������ںϽ����Ĵ��£�����������һ���������÷�Ӧ�Ļ�ѧ����ʽΪ__________________�����з�Ӧ�еİ��백�������еİ�������ͬ����________________��
A.2Na+2NH3=2NaNH2+H2��
B.2NH3+3CuO=3Cu+N2+3H2O
C.4NH3+6NO=5N2+6H2O
D.HNO3+NH3=NH4NO3
���𰸡� 22.3 500L 4NH3+5O2![]() 4NO+6H2O BC
4NO+6H2O BC
��������(1)500L���������ʵ���Ϊ�� ![]() =22.32mol������ˮ�γ�1L��ˮ��c=
=22.32mol������ˮ�γ�1L��ˮ��c=![]() =
=![]() =22.3mol/L�����������백�������ʵ�����Ӧ������������泥��ð�ˮ��������ն��������ڱ�״���µ�����백���������ȣ�Ϊ500L���ʴ�Ϊ��22.3��500L��
=22.3mol/L�����������백�������ʵ�����Ӧ������������泥��ð�ˮ��������ն��������ڱ�״���µ�����백���������ȣ�Ϊ500L���ʴ�Ϊ��22.3��500L��
(2)�����Ϳ����Ļ����ͨ�����ȵIJ���Ͻ���������������һ����������Ӧ�Ļ�ѧ����ʽΪ4NH3+5O2![]() 4NO+6H2O����Ӧ�а����ǻ�ԭ����A. 2Na+2NH3 = 2NaNH2+H2����Ӧ�а�����������������B. 2NH3+3CuO = 3Cu+N2+3H2O ��Ӧ�а����ǻ�ԭ������ȷ��
4NO+6H2O����Ӧ�а����ǻ�ԭ����A. 2Na+2NH3 = 2NaNH2+H2����Ӧ�а�����������������B. 2NH3+3CuO = 3Cu+N2+3H2O ��Ӧ�а����ǻ�ԭ������ȷ��
C. 4NH3+6NO = 5N2+6H2O��Ӧ�а����ǻ�ԭ������ȷ��D. HNO3+NH3 = NH4NO3��Ӧ�а����Ȳ���������Ҳ���ǻ�ԭ��������ѡBC���ʴ�Ϊ��4NH3+5O2![]() 4NO+6H2O��BC��
4NO+6H2O��BC��

����Ŀ���±������ڱ��е�һ���֣�����A��I�����ڱ��е�λ�ã���Ԫ�ط��Ż�ѧʽ�ش��������⣺
| ��A | ��A | ��A | ��A | ��A | ��A | ��A | 0 |
1 | A | |||||||
2 | D | E | G | I | ||||
3 | B | C | F | H |
��1����Ԫ�أ���ѧ��������õ��� ��ֻ�и��۶������۵��� ����������ǿ�ĵ����� ����ԭ����ǿ�ĵ����� ��
��2������������ˮ�����м�����ǿ���� ��������ǿ���� �������Ե��� ��
��3��A�ֱ���D��E��F��G��H�γɵĻ������У����ȶ��� ��
��4����B��C��D��E��F��G��H�У�ԭ�Ӱ뾶������ .��С���� ��