��Ŀ����
����Ŀ��(1)˳-1��2-����������ͷ�-1��2-����������ɷ�������ת����
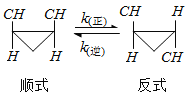
�÷�Ӧ�����ʷ��̿ɱ�ʾΪ��v(��)=k(��)c(˳)��v(��)=k(��)c(��)��k(��)��k(��)��һ���¶�ʱΪ�������ֱ���������淴Ӧ���ʳ������ش��������⣺
��֪��t1�¶��£�k(��)=0.006s��1��k(��)=0.002s��1�����¶��·�Ӧ��ƽ��ʱ��c(��-1��2-����������)��c(˳-1��2-����������)=_____________��
�÷�Ӧ�Ļ��Ea(��)С��Ea(��)����![]() ________0(����С����������������������)��
________0(��������������������������)��
(2)����ԭCO2�ǽ������ЧӦ����Դ�������Ҫ�ֶ�֮һ���о���������Cu/ZnO���������£�CO2��H2�ɷ���������Ӧ���ֱ�����CH3OH��CO����Ӧ���Ȼ�ѧ����ʽ���£�
CO2(g)+3 H2(g) CH3OH(g)+H2O(g)��H1=-53.7kJ��mol-1 I
CO2(g)+ H2(g) CO(g)+H2O(g)��H2 II
ijʵ���ҿ���CO2��H2��ʼͶ�ϱ�Ϊ1:2.2��������ͬ��Ӧʱ��������ʵ�����ݣ�
T��K�� | ���� | CO2ת���ʣ�%�� | �״�ѡ���ԣ�%�� |
543 | Cat.1 | 12.3 | 42.3 |
543 | Cat.2 | 10.9 | 72.7 |
553 | Cat.1 | 15.3 | 39.1 |
553 | Cat.2 | 12.0 | 71.6 |
����ע��Cat.1:Cu/ZnO���װ���Cat.2:Cu/ZnO����Ƭ���״�ѡ���ԣ�ת����CO2�����ɼ�ȩ�İٷֱȣ�
��֪����CO��H2��ȼ���ȷֱ�Ϊ-283.0kJ��mol-1��-285.8kJ��mol-1
��H2O(l)=H2O(g) ��H3=44.0kJ��mol-1
��ش�(�������¶ȶ���H��Ӱ��)����ӦII����H2= kJ��mol-1��
����ͼ�зֱ�I����������Cat.1����Cat.2�������������Ӧ����-������ʾ��ͼ(��ͼ�б�ע����Ӧ������)��

���𰸡�
(1)3����(2)+41.2��
 ��
��
��������
���������(1)����v(��)=k(��)c(˳)��k(��)=0.006s-1����v(��)=0.006c(˳)��v(��)=k(��)c(��)��k(��)=0.002s-1����v(��)=0.002c(��)����ѧƽ��״̬ʱ���淴Ӧ������ȣ���0.006c(˳)=0.002c(��)�����¶��·�Ӧ��ƽ�ⳣ��ֵK1=![]() =
=![]() =3���÷�Ӧ�Ļ��Ea(��)С��Ea(��)��˵���ϼ����յ�����С�ڳɼ��ͷŵ����������÷�ӦΪ���ȷ�Ӧ������HС��0���ʴ�Ϊ��3��С�ڣ�
=3���÷�Ӧ�Ļ��Ea(��)С��Ea(��)��˵���ϼ����յ�����С�ڳɼ��ͷŵ����������÷�ӦΪ���ȷ�Ӧ������HС��0���ʴ�Ϊ��3��С�ڣ�
(2)��֪��CO��H2�ı�ȼ���ȷֱ�Ϊ-283.0kJmol-1��-285.8kJmol-1����H2O(1)�TH2O(g)��H3=44.0kJmol-1����֪�Ȼ�ѧ����ʽa��CO(g)+![]() O2(g)=CO2(g)��H=-283.0kJmol-1��b��H2(g)+
O2(g)=CO2(g)��H=-283.0kJmol-1��b��H2(g)+![]() O2(g)=H2O(1)��H=-285.8kJmol-1��c��H2O(1)�TH2O(g)��H3=44.0kJmol-1���ɸ�˹���ɽ�b-a+c�ɵ�CO2(g)+H2(g)CO(g)+H2O(g)��H2 =(-285.8+283.0+44)kJmol-1=+41.2kJmol-1���ӱ������ݷ������ڴ���Cat.2�������£��״���ѡ���Ը���˵������Cat.2�Է�Ӧ����Ч�����ã������ܽ��ͷ�Ӧ�Ļ�ܣ�˵��ʹ�ô���Cat.2�ķ�Ӧ�����л�ܸ��ͣ���ͼΪ
O2(g)=H2O(1)��H=-285.8kJmol-1��c��H2O(1)�TH2O(g)��H3=44.0kJmol-1���ɸ�˹���ɽ�b-a+c�ɵ�CO2(g)+H2(g)CO(g)+H2O(g)��H2 =(-285.8+283.0+44)kJmol-1=+41.2kJmol-1���ӱ������ݷ������ڴ���Cat.2�������£��״���ѡ���Ը���˵������Cat.2�Է�Ӧ����Ч�����ã������ܽ��ͷ�Ӧ�Ļ�ܣ�˵��ʹ�ô���Cat.2�ķ�Ӧ�����л�ܸ��ͣ���ͼΪ ���ʴ�Ϊ��+41.2��
���ʴ�Ϊ��+41.2�� ��
��