��Ŀ����
����Ŀ���ҹ���������Ԥ����������Ҫ�����������棺�������������NOx������������������ִ�����Ⱦ���Ũ�ȣ�
��1�����뵪�������йص�ȫ��������Դ�������������������ĸ��ţ���
a������ b��ɳ���� c���⻯ѧ���� d������ЧӦ
��������������ø�ѹ�������������������
��2��Ϊ�˽�������β���Դ�������Ⱦ��Ŀǰ����Ч�ķ����Ǹ�������װβ������װ�ã����ܽ�β���е�һ����̼��NO�ڴ��������£�������Ӧת��Ϊ�����壬�䷴Ӧ�Ļ�ѧ����ʽΪ ��
��3����д��ʵ�������ɹ���������ȡ�����Ļ�ѧ����ʽ��
�����ռ�һƿ����İ�����ѡ��ͼ1�е�װ�ã�������˳��Ϊ������װ�á���������������Сд��ĸ��ʾ��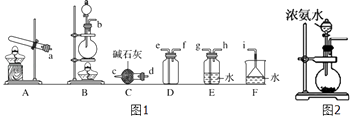
��ʵ����Ҳ����ͼ2��ʾװ����ȡ��������ƿ�ڹ����ѡ����ѡ��ѡ��Ĵ��ţ���
a����ʯ�� b����ʯ�� c���������� d������������
�ܴ��������£�NH3Ҳ����������NOx����Ⱦ���������ֶԻ����������ʣ���д��NH3��NO2��Ӧ�Ļ�ѧ����ʽΪ �� �÷�Ӧ��ÿ����0.5molN2ת�� mol���ӣ�
���𰸡�
��1��ac����Ӿ
��2��2NO+2CO ![]() N2+2CO2
N2+2CO2
��3��2NH4Cl+Ca��OH��2 ![]() CaCl2+2NH3��+2H2O��d��c��f��e��i��ab��6NO2+8NH3
CaCl2+2NH3��+2H2O��d��c��f��e��i��ab��6NO2+8NH3 ![]() 7N2+12H2O��1.7
7N2+12H2O��1.7
���������⣺��1����a��������������ˮ�������ᣮʹ��ˮpH��5.6����aѡ��
b��ɳ��������������������йأ��뵪�������أ���b��ѡ��
c���⻯ѧ����������β���еĵ����������йأ���cѡ��
d����ɫ��Ⱦ�������йأ���d��ѡ��
ͨ�����Ϸ���֪���뵪�������йصĻ�����Ⱦ�����ꡢ�⻯ѧ������
���Դ��ǣ�ac��
�ڽ�����е�Ӿ�����ʣ�����ӵ�Դ�����£�����Ľ������ܹ����������ƶ����������ø�ѹ������������ڼ���������Ԥ��������Ⱦ�������ý����Ӿ�����ʣ�����������������ø�ѹ������������˵�Ӿ����
���Դ��ǣ���Ӿ����2��NO��CO�ڴ���������ת��Ϊ�����Ͷ�����̼����Ӧ�ķ���ʽΪ��2NO+2CO ![]() N2+2CO2 ��
N2+2CO2 ��
���Դ��ǣ�2NO+2CO ![]() N2+2CO2����3����ʵ�������ɹ���������ȡ����Ϊ�����Ȼ�狀����������Ʒ�Ӧ����Ӧ�ķ���ʽΪ��Ca��OH��2+2NH4Cl
N2+2CO2����3����ʵ�������ɹ���������ȡ����Ϊ�����Ȼ�狀����������Ʒ�Ӧ����Ӧ�ķ���ʽΪ��Ca��OH��2+2NH4Cl ![]() CaCl2+2NH3��+2H2O��
CaCl2+2NH3��+2H2O��
���Դ��ǣ�Ca��OH��2+2NH4Cl ![]() CaCl2+2NH3��+2H2O��
CaCl2+2NH3��+2H2O��
��ʵ�����ü��ȹ����Ȼ�狀��������Ƶķ����Ʊ��������Ʊ��������к���ˮ����������Ϊ�������壬Ӧѡ��ʢ�м�ʯ�Ҹ���ܸ������壬������������ˮ���ܶ�С�ڿ����ܶ�����Ӧѡ�������ſ������ռ����壬������������ˮ��β��������ˮ���գ�ע���ֹ�����ķ�����������ȷ������˳��Ϊ������װ�á�d��c��f��e��i��
���Դ��ǣ�d��c��f��e��i��
��a����ʯ���еμӰ�ˮ����ʯ���ܽ���ȣ�ʹ�����ų�������ȷ��
b����ʯ���е��백ˮ���ܽ���ȣ�ʹ�ų�����������ȷ��
C����������������ˮ�����ܷų���������ˮ��Ӱ�죬�������ɰ������ʴ���
D�����������Ͱ�ˮ��Ӧ�����������Σ��������ɰ������ʴ���
���Դ��ǣ�ab��
��NH3����������NO����Ⱦ������2�ֶԻ����������ʣ�Ӧ���ɵ�����ˮ���䷴Ӧ����ʽΪ6NO2+8NH3 ![]() 7N2+12H2O���ɷ���ʽ��֪������7molN2 �� ת��24mol���ӣ���ÿ����0.5molN2ת��1.7mol���ӣ�
7N2+12H2O���ɷ���ʽ��֪������7molN2 �� ת��24mol���ӣ���ÿ����0.5molN2ת��1.7mol���ӣ�
���Դ��ǣ�6NO2+8NH3 ![]() 7N2+12H2O��1.7��
7N2+12H2O��1.7��

 ������������Ӧ����ϵ�д�
������������Ӧ����ϵ�д� ͬ����չ�Ķ�ϵ�д�
ͬ����չ�Ķ�ϵ�д�