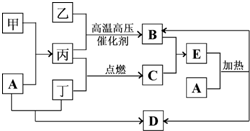
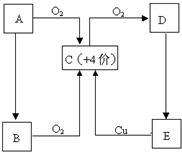
��Ŀ����
��ͼ���ɶ�����Ԫ����ɵ�һЩ���ʼ��仯����֮���ת����ϵͼ�����³�ѹ�£�D��F��K��Ϊ��ɫ�̼�����ζ�����壬����K����������ЧӦ��B���������ɫҺ�壬A���ɵ���C��D��ȼ�����ɵĵ���ɫ���壮����Ӧ�����ɵIJ�����������ȥ����ش��������⣺

��1������A�Ļ�ѧʽΪ______��
��2��K�ĵ���ʽΪ______��
��3����Ӧ�ٵ����ӷ���ʽΪ______����Ӧ�ڵĻ�ѧ����ʽΪ______��

��1������A�Ļ�ѧʽΪ______��
��2��K�ĵ���ʽΪ______��
��3����Ӧ�ٵ����ӷ���ʽΪ______����Ӧ�ڵĻ�ѧ����ʽΪ______��
A���ɵ���C��D��ȼ�����ɵĵ���ɫ���壬ӦΪNa2O2��B���������ɫҺ�壬ӦΪH2O����DΪO2��EΪNaOH��CΪNa��FΪH2����ת����ϵ��֪KΪCO2��HΪNa2CO3��IΪNaHCO3��
��1�������Ϸ�����֪AΪNa2O2���ʴ�Ϊ��Na2O2��
��2�������Ϸ�����֪������KΪCO2������ʽΪ ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
��3����Ӧ��ΪNa��H2O�ķ�Ӧ����Ӧ�����ӷ���ʽΪ2Na+2H2O�T2Na++2OH-+H2������Ӧ��Ϊ2NaHCO3
Na2CO3+CO2��+H2O��
�ʴ�Ϊ��2Na+2H2O�T2Na++2OH-+H2����2NaHCO3
Na2CO3+CO2��+H2O��
��1�������Ϸ�����֪AΪNa2O2���ʴ�Ϊ��Na2O2��
��2�������Ϸ�����֪������KΪCO2������ʽΪ
 ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
����3����Ӧ��ΪNa��H2O�ķ�Ӧ����Ӧ�����ӷ���ʽΪ2Na+2H2O�T2Na++2OH-+H2������Ӧ��Ϊ2NaHCO3
| ||
�ʴ�Ϊ��2Na+2H2O�T2Na++2OH-+H2����2NaHCO3
| ||

��ϰ��ϵ�д�
�����Ŀ
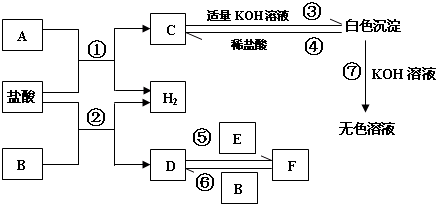

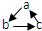 ����������ʾһ����ɣ���ϵת�����ǣ�������
����������ʾһ����ɣ���ϵת�����ǣ�������