��Ŀ����
7��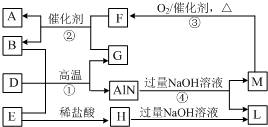 AlN��һ�������½ṹ�մɣ���ҵ�Ͽ���ͼ�еķ�Ӧ���Ʊ�����Ӧ�ڳ����ڴ�������β����B��D�ǵ��ʣ�A��E��F��G�������A���γ�����ЧӦ�����壬M��ʹʪ��ĺ�ɫʯ����ֽ���������壨ת�������еIJ��ֲ�������ȥ������ش��������⣺
AlN��һ�������½ṹ�մɣ���ҵ�Ͽ���ͼ�еķ�Ӧ���Ʊ�����Ӧ�ڳ����ڴ�������β����B��D�ǵ��ʣ�A��E��F��G�������A���γ�����ЧӦ�����壬M��ʹʪ��ĺ�ɫʯ����ֽ���������壨ת�������еIJ��ֲ�������ȥ������ش��������⣺��1��A�ĵ���ʽΪ
 ��
����2��G�Ļ�ѧʽΪCO��
��3����Ӧ�۵Ļ�ѧ����ʽΪ4NH3+5O2$\frac{\underline{����}}{��}$4NO+6H2O��
��4����Ӧ�ܵ����ӷ���ʽΪAlN+OH-+H2O=AlO2-+NH3����
���� M����ʹʪ��ĺ�ɫʯ����ֽ���������壬��MΪNH3���ɷ�Ӧ�ܿ�֪LΪNaAlO2��M��������Ӧ����F���ҷ�Ӧ�ڳ����ڴ�������β����A���γ�����ЧӦ�����壬��FΪNO��AΪCO2��������GΪCO������BΪN2������֪����DΪC��������EΪAl2O3��HΪAlCl3���ݴ˽��
��� �⣺M����ʹʪ��ĺ�ɫʯ����ֽ���������壬��MΪNH3���ɷ�Ӧ�ܿ�֪LΪNaAlO2��M��������Ӧ����F���ҷ�Ӧ�ڳ����ڴ�������β����A���γ�����ЧӦ�����壬��FΪNO��AΪCO2��������GΪCO������BΪN2������֪����DΪC��������EΪAl2O3��HΪAlCl3��
��1�������Ϸ�����֪AΪCO2������ʽΪ ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
��2��������������֪��GΪCO���ʴ�Ϊ��CO��
��3����Ӧ��Ϊ�����Ĵ���������Ӧ�Ļ�ѧ����ʽΪ4NH3+5O2$\frac{\underline{����}}{��}$4NO+6H2O��
�ʴ�Ϊ��4NH3+5O2$\frac{\underline{����}}{��}$4NO+6H2O��
��4����Ӧ�ܵ����ӷ���ʽΪ��AlN+OH-+H2O=AlO2-+NH3����
�ʴ�Ϊ��AlN+OH-+H2O=AlO2-+NH3����
���� ���⿼�������ƶϣ���M�����ʡ�AΪ����ЧӦ���塱���ƶ�ͻ�ƿڣ����ת����ϵ�ƶϣ�ͬʱ����ѧ������˼ά��������ȷ���ʵ������ǽⱾ��Ĺؼ����Ѷ��еȣ�

| A�� | 12C��14C | B�� | ˮ�ͱ� | C�� | O2��O3 | D�� | CO��CO2 |
| A�� | һ��������Ba2+��NH4+���ܴ��� | B�� | CO32- һ������ | ||
| C�� | Na+һ������ | D�� | һ��������Cl- |
| A�� | 2H++2e-�TH2 | B�� | Fe-2e-�TFe2+ | ||
| C�� | 2H2O+O2+4e-�T4OH- | D�� | Fe3++e-�TFe2+ |
| A�� | ������ԭ�Ӽ�����γɻ�ѧ�� | |
| B�� | �������ǽ���ԭ���γɵĻ������в����ܴ������Ӽ� | |
| C�� | �κλ�ѧ���ı仯��Ȼ�����ŵ��ӵĵ�ʧ | |
| D�� | ij�������ˮ��Һ�ܵ��磬��û�������һ���������Ӽ� |



 ��
�� ��
�� ��
��