��Ŀ����
��14�֣���ѧ����Ϊ����������˷������ʻ���������Ϊ�������ɳ�����չ�����˾��ס�
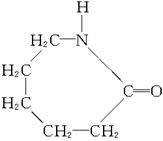
��1���������������Ļ��������ճ���ʳ����Ҫ��ɲ��֡�����ʳ��ٻƹ�
�ڴ� �۵��� �ܼ��� �����Ѹɣ����и��������ʵ���
������ţ���
��2�����������������������зdz���Ҫ�IJ��ϣ���ÿ������ʴ����ʧ������ʮ�־��ˣ���ɸ�����ʴ����Ҫԭ���Ƿ����绯ѧ��ʴ���ڵ绯ѧ��ʴ�У�ˮĤ���Խ����������ʱ������������ʴ����缫��ӦΪ������:O2+2H2O+4e-=4OH-;������ ��ˮĤ���Խ�ǿʱ���������ⸯʴ����缫��ӦΪ������:________________ _ ��
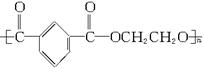
��3�������е�ˮ�ࡢ��ͨ�������մɶ����� ����������ϡ������ǽ������ϡ��͡��߷��Ӻϳɲ��ϡ�����������ԭ�ϲ���Ҫʹ��ʯ��ʯ���� �����ϳ�����Ϊһ��ʱ���ı�־��������ʱ������ͭ��ʱ��������ʱ��������Ϣʱ��������Ϊ��Ϣ�����Ĺؼ����ϵĵĻ�ѧʽ�� ��
(4) ̼�������dz��������ɼ��Ϳ���ҩ����д��������ҩʱ�����ӷ���ʽ
.
��1���������������Ļ��������ճ���ʳ����Ҫ��ɲ��֡�����ʳ��ٻƹ�
�ڴ� �۵��� �ܼ��� �����Ѹɣ����и��������ʵ���
������ţ���
��2�����������������������зdz���Ҫ�IJ��ϣ���ÿ������ʴ����ʧ������ʮ�־��ˣ���ɸ�����ʴ����Ҫԭ���Ƿ����绯ѧ��ʴ���ڵ绯ѧ��ʴ�У�ˮĤ���Խ����������ʱ������������ʴ����缫��ӦΪ������:O2+2H2O+4e-=4OH-;������ ��ˮĤ���Խ�ǿʱ���������ⸯʴ����缫��ӦΪ������:________________ _ ��
��3�������е�ˮ�ࡢ��ͨ�������մɶ����� ����������ϡ������ǽ������ϡ��͡��߷��Ӻϳɲ��ϡ�����������ԭ�ϲ���Ҫʹ��ʯ��ʯ���� �����ϳ�����Ϊһ��ʱ���ı�־��������ʱ������ͭ��ʱ��������ʱ��������Ϣʱ��������Ϊ��Ϣ�����Ĺؼ����ϵĵĻ�ѧʽ�� ��
(4) ̼�������dz��������ɼ��Ϳ���ҩ����д��������ҩʱ�����ӷ���ʽ
.
(1)���ڢ� (2) Fe �C 2e- = Fe2+ 2H+ + 2e- = H2��
(3) ���ǽ������� �մ� SiO2
(4) HCO3- + H+ = H2O + CO2��
(3) ���ǽ������� �մ� SiO2
(4) HCO3- + H+ = H2O + CO2��
(1)�������и��������ʣ��ƹϸ���ά���أ������������࣮���ڢ� (2)����ʧȥ���ӱ������� Fe �C 2e- = Fe2+ �����õ��ӷ�����ԭ��Ӧ��2H+ + 2e- = H2��
(3) ���ǽ�����������ijЩԪ�ص������̼��������±�ػ���������Լ������Ρ������Ρ������Ρ������ε�������ɵIJ��ϡ����ǽ������� �մ� SiO2
(4) HCO3- + H+ = H2O + CO2��

��ϰ��ϵ�д�
 ������ȫ��������ϵ�д�
������ȫ��������ϵ�д�
�����Ŀ
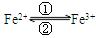 ��ת��ʱ�����е�Fe2+�� �������е�Fe3+�� ��
��ת��ʱ�����е�Fe2+�� �������е�Fe3+�� ��