��Ŀ����
��10�֣�ƻ��ζ������һ����ƻ�����Ͷ��ɵ�������Ʒ�����нⶾ����֬��Ч��������Ҫ��������Ϊƻ���ᡣƻ�����ڷ����ᴿ��Ļ�ѧ�������£�
����Է�������������150����ȫȼ�պ�ֻ����CO2��H2O��������C��H�����������ֱ�Ϊ35.82%��4.48%����1mol������������NaHCO3��Ӧ�ų�44.8L CO2����������Na��Ӧ�ų�H2 33.6L�����������Ϊ��������ۺ˴ż�⣬�÷����д������ֻ�ѧ������ͬ��̼ԭ�ӣ���ԭ��Ҳ�������ֲ�ͬ�Ļ�ѧ��������ش�
��1���������� ���Ȼ���ƻ�������ʽΪ ��
��2��ƻ����Ľṹ��ʽΪ������������������������������ ��
��3��ƻ�����ͬ���칹���з��������١���������������
��д����һ�ֵĽṹ��ʽ����
��4��������һ�������¿��Է�����������ˮ���仯ѧ����ʽΪ�� ��
����Է�������������150����ȫȼ�պ�ֻ����CO2��H2O��������C��H�����������ֱ�Ϊ35.82%��4.48%����1mol������������NaHCO3��Ӧ�ų�44.8L CO2����������Na��Ӧ�ų�H2 33.6L�����������Ϊ��������ۺ˴ż�⣬�÷����д������ֻ�ѧ������ͬ��̼ԭ�ӣ���ԭ��Ҳ�������ֲ�ͬ�Ļ�ѧ��������ش�
��1���������� ���Ȼ���ƻ�������ʽΪ ��
��2��ƻ����Ľṹ��ʽΪ������������������������������ ��
��3��ƻ�����ͬ���칹���з��������١���������������
��д����һ�ֵĽṹ��ʽ����
��4��������һ�������¿��Է�����������ˮ���仯ѧ����ʽΪ�� ��
��1��2�� C4H6O5
��2��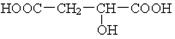 ��3��
��3�� 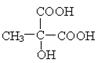
��4��

��2��
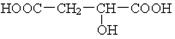 ��3��
��3�� 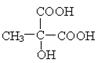
��4��

�����л������ʽ���ṹʽ���жϡ����ݢڿ�֪�����к���2���Ȼ���1���ǻ������ݢۿ�֪���������ٺ���4��̼ԭ�ӡ����ݢٿ�֪����̼ԭ�ӵĸ��������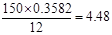 �����Է����о�ֻ�ܺ���4��̼ԭ�ӣ������Է���������
�����Է����о�ֻ�ܺ���4��̼ԭ�ӣ������Է���������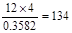 ������ԭ�Ӹ�����
������ԭ�Ӹ�����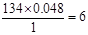 �������ԭ�ӵĸ�����
�������ԭ�ӵĸ�����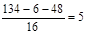 ��������ʽΪC4H6O5����Ϊ��ԭ��Ҳ�������ֲ�ͬ�Ļ�ѧ������������ṹ��ʽΪ
��������ʽΪC4H6O5����Ϊ��ԭ��Ҳ�������ֲ�ͬ�Ļ�ѧ������������ṹ��ʽΪ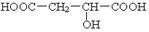 �������ǻ�̼ԭ�ӵ���λ̼ԭ�Ӻ�����ԭ�ӣ����Կɷ�����ȥ��Ӧ����̼̼˫��������٢ڵ�ͬ���칹�������
�������ǻ�̼ԭ�ӵ���λ̼ԭ�Ӻ�����ԭ�ӣ����Կɷ�����ȥ��Ӧ����̼̼˫��������٢ڵ�ͬ���칹�������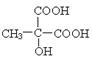 ��
��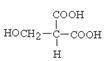 ��
��
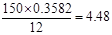 �����Է����о�ֻ�ܺ���4��̼ԭ�ӣ������Է���������
�����Է����о�ֻ�ܺ���4��̼ԭ�ӣ������Է���������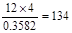 ������ԭ�Ӹ�����
������ԭ�Ӹ�����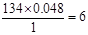 �������ԭ�ӵĸ�����
�������ԭ�ӵĸ�����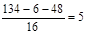 ��������ʽΪC4H6O5����Ϊ��ԭ��Ҳ�������ֲ�ͬ�Ļ�ѧ������������ṹ��ʽΪ
��������ʽΪC4H6O5����Ϊ��ԭ��Ҳ�������ֲ�ͬ�Ļ�ѧ������������ṹ��ʽΪ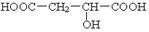 �������ǻ�̼ԭ�ӵ���λ̼ԭ�Ӻ�����ԭ�ӣ����Կɷ�����ȥ��Ӧ����̼̼˫��������٢ڵ�ͬ���칹�������
�������ǻ�̼ԭ�ӵ���λ̼ԭ�Ӻ�����ԭ�ӣ����Կɷ�����ȥ��Ӧ����̼̼˫��������٢ڵ�ͬ���칹�������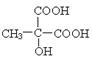 ��
�� ��
��
��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
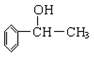
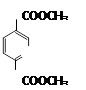 ��ͬ���칹���ж��֣��������������Ĺ��� �֡�
��ͬ���칹���ж��֣��������������Ĺ��� �֡�