��Ŀ����
��2012?����һģ��FeSO4?7H2O�㷺����ҽҩ��ҵ����������FeSO4?7H2O��ʵ�����Ʊ�����ͼ�������������������գ�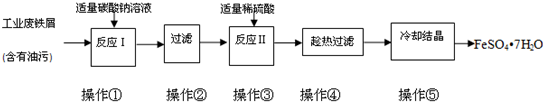
��1����������̼������Һ�ܳ�ȥ���ۣ�ԭ����
��2���������ʷ��뷽����ԭ���������ͬ����
a������ b����Һ
c������ d���ؽᾧ
��3��FeSO4?7H2O��ijЩ��Ѫ������Ҫ�ɷ֣�ʵ������Ϊ�ⶨij��Ѫ������Ԫ�صĺ���������������ʵ�飺
������100mL 1.00��10-2mol?L-1��KMnO4��Һ�����õ���������ƽ���ձ�����ͷ�ιܼ�
a��KMnO4����ˮ�����ȣ�����ֱ��������ƿ���ܽ�
b������ƿϴ�Ӻ�����T��ֱ������ʵ��
c�����ݺ�ҡ�ȣ���Һ����ڿ̶��ߣ��ټ�ˮ����Һ����͵���̶�����ƽ
d���������ʱ��ˮ�����̶��߱�����������
��ȡ2.0g�˸ò�Ѫ�������Һ������Ԥ������ʹ���е���Ԫ��ȫ����Fe2+���������ƺõı�KMnO4��Һ�����������½���������ԭ�ζ�����Ӧ�����ӷ���ʽ�ǣ�5Fe2++MnO4-+8H+�T5Fe3++Mn2++4H2O�����еζ�ʱ����KMnO4��ҺӦ��ʢ����
�۵ζ�����ʱ������Ũ��Ϊ1.00��10-2mol?L-1�ı�KMnO4��Һ26.00mL����ò�Ѫ������Ԫ�صĺ���Ϊ

��1����������̼������Һ�ܳ�ȥ���ۣ�ԭ����
CO32-+H2O?HCO3-+OH-��HCO3-+H2O?H2CO3+OH-
CO32-+H2O?HCO3-+OH-��HCO3-+H2O?H2CO3+OH-
�������ӷ���ʽ��ʾ������2���������ʷ��뷽����ԭ���������ͬ����
c
c
���������ĸ����a������ b����Һ
c������ d���ؽᾧ
��3��FeSO4?7H2O��ijЩ��Ѫ������Ҫ�ɷ֣�ʵ������Ϊ�ⶨij��Ѫ������Ԫ�صĺ���������������ʵ�飺
������100mL 1.00��10-2mol?L-1��KMnO4��Һ�����õ���������ƽ���ձ�����ͷ�ιܼ�
100mL����ƿ��������
100mL����ƿ��������
�����ƹ���������˵���У���ȷ����bd
bd
���������ĸ����a��KMnO4����ˮ�����ȣ�����ֱ��������ƿ���ܽ�
b������ƿϴ�Ӻ�����T��ֱ������ʵ��
c�����ݺ�ҡ�ȣ���Һ����ڿ̶��ߣ��ټ�ˮ����Һ����͵���̶�����ƽ
d���������ʱ��ˮ�����̶��߱�����������
��ȡ2.0g�˸ò�Ѫ�������Һ������Ԥ������ʹ���е���Ԫ��ȫ����Fe2+���������ƺõı�KMnO4��Һ�����������½���������ԭ�ζ�����Ӧ�����ӷ���ʽ�ǣ�5Fe2++MnO4-+8H+�T5Fe3++Mn2++4H2O�����еζ�ʱ����KMnO4��ҺӦ��ʢ����
��ʽ
��ʽ
�����ʽ����ʽ��֮һ���ζ����У�ԭ����KMnO4��Һ��ǿ�������ܸ�ʴ��
KMnO4��Һ��ǿ�������ܸ�ʴ��
���۵ζ�����ʱ������Ũ��Ϊ1.00��10-2mol?L-1�ı�KMnO4��Һ26.00mL����ò�Ѫ������Ԫ�صĺ���Ϊ
3.64
3.64
%����������1��̼������Һ�ܳ�ȥ��������Ϊ̼�������ˮ��Һ��ˮ���Լ��ԣ�
��2��������������ֽ�ѹ��岻�������Һ���룻�����Է����жϣ�
��3��������������Һ�Ĺ��̺Ͳ���ѡ��������������Һ���Ƶ�ʵ������жϣ�
a������ƿ���������ܽ����ʣ�
b������ƿ��ȷ��������Һ�����ߣ�����ˮ���ݣ�
c�����ݺ�ҡ�Ⱥ���Ҫ�ټ�ˮ����ˮ����ҺŨ�ȼ�С��
d����ˮ�����̶ȣ�����ʧ����Ҫ�������ƣ�
��KMnO4��Һ����ǿ�����ԣ�����ʢ���ڼ�ʽ�ζ��ܣ���Ҫѡ����ʽ�ζ��ܣ�
������������ԭ��Ӧ�����ӷ���ʽ����õ���
��2��������������ֽ�ѹ��岻�������Һ���룻�����Է����жϣ�
��3��������������Һ�Ĺ��̺Ͳ���ѡ��������������Һ���Ƶ�ʵ������жϣ�
a������ƿ���������ܽ����ʣ�
b������ƿ��ȷ��������Һ�����ߣ�����ˮ���ݣ�
c�����ݺ�ҡ�Ⱥ���Ҫ�ټ�ˮ����ˮ����ҺŨ�ȼ�С��
d����ˮ�����̶ȣ�����ʧ����Ҫ�������ƣ�
��KMnO4��Һ����ǿ�����ԣ�����ʢ���ڼ�ʽ�ζ��ܣ���Ҫѡ����ʽ�ζ��ܣ�
������������ԭ��Ӧ�����ӷ���ʽ����õ���
����⣺��1����������̼������Һ�ܳ�ȥ���ۣ���Һ��̼�������ˮ��Һ��ˮ���Լ��ԣ���Ӧ�����ӷ���ʽΪ��CO32-+H2O?HCO3-+OH-��HCO3-+H2O?H2CO3+OH-��
�ʴ�Ϊ��CO32-+H2O?HCO3-+OH-��HCO3-+H2O?H2CO3+OH-��
��2�����ʷ��뷽����ԭ���������ͬ����������ֽ�ѹ��岻�������Һ���룻�����Է����жϣ�
a�������Ƿ���е㲻ͬ�Ļ�����a�����ϣ�
b����Һ�����û������ܵ�Һ�徲�÷�Һ����b�����ϣ�
c�����������ð�Ĥ������Һ�ͽ��壬����ԭ����ͬ����c���ϣ�
d���ؽᾧ����������Ũ�����ᾧ��������d�����ϣ�
�ʴ�Ϊ��c��
��3��������100mL 1.00��10-2mol?L-1��KMnO4��Һ�����õ���������ƽ���ձ�����ͷ�ιܡ�100mL����ƿ ��������
a������ƿ���������ܽ����ʣ���a����
b������ƿ��ȷ��������Һ�����ߣ�����ˮ���ݣ�����ƿϴ�Ӻ�����T��ֱ������ʵ�飻��b��ȷ��
c�����ݺ�ҡ�Ⱥ���Ҫ�ټ�ˮ����ˮ����ҺŨ�ȼ�С����c����
d����ˮ�����̶ȣ�����ʧ����Ҫ�������ƣ���d��ȷ��
�ʴ�Ϊ��100mL����ƿ����������bd��
��KMnO4��Һ����ǿ�����ԣ�����ʢ���ڼ�ʽ�ζ��ܣ���Ϊ���������Һ��ʴ�ܣ�����ʢ������ʽ�ζ����У�
�ʴ�Ϊ����ʽ��KMnO4��Һ��ǿ�������ܸ�ʴ�ܣ�
�۵ζ�����ʱ������Ũ��Ϊ1.00��10-2mol?L-1�ı�KMnO4��Һ26.00mL����ʴ�ķ�ӦΪ������Ԫ�����ʵ���Ϊx��
5Fe2++MnO4-+8H+�T5Fe3++Mn2++4H2O��
5 1
x 1.00��10-2mol?L-1��0.0260L
x=1.3��10-3mol��
��Ԫ������=1.3��10-3mol��56g/mol=0.0728g
2.0g�˸ò�Ѫ������Ԫ�غ���=
��100%=3.64%
�ʴ�Ϊ��3.64��
�ʴ�Ϊ��CO32-+H2O?HCO3-+OH-��HCO3-+H2O?H2CO3+OH-��
��2�����ʷ��뷽����ԭ���������ͬ����������ֽ�ѹ��岻�������Һ���룻�����Է����жϣ�
a�������Ƿ���е㲻ͬ�Ļ�����a�����ϣ�
b����Һ�����û������ܵ�Һ�徲�÷�Һ����b�����ϣ�
c�����������ð�Ĥ������Һ�ͽ��壬����ԭ����ͬ����c���ϣ�
d���ؽᾧ����������Ũ�����ᾧ��������d�����ϣ�
�ʴ�Ϊ��c��
��3��������100mL 1.00��10-2mol?L-1��KMnO4��Һ�����õ���������ƽ���ձ�����ͷ�ιܡ�100mL����ƿ ��������
a������ƿ���������ܽ����ʣ���a����
b������ƿ��ȷ��������Һ�����ߣ�����ˮ���ݣ�����ƿϴ�Ӻ�����T��ֱ������ʵ�飻��b��ȷ��
c�����ݺ�ҡ�Ⱥ���Ҫ�ټ�ˮ����ˮ����ҺŨ�ȼ�С����c����
d����ˮ�����̶ȣ�����ʧ����Ҫ�������ƣ���d��ȷ��
�ʴ�Ϊ��100mL����ƿ����������bd��
��KMnO4��Һ����ǿ�����ԣ�����ʢ���ڼ�ʽ�ζ��ܣ���Ϊ���������Һ��ʴ�ܣ�����ʢ������ʽ�ζ����У�
�ʴ�Ϊ����ʽ��KMnO4��Һ��ǿ�������ܸ�ʴ�ܣ�
�۵ζ�����ʱ������Ũ��Ϊ1.00��10-2mol?L-1�ı�KMnO4��Һ26.00mL����ʴ�ķ�ӦΪ������Ԫ�����ʵ���Ϊx��
5Fe2++MnO4-+8H+�T5Fe3++Mn2++4H2O��
5 1
x 1.00��10-2mol?L-1��0.0260L
x=1.3��10-3mol��
��Ԫ������=1.3��10-3mol��56g/mol=0.0728g
2.0g�˸ò�Ѫ������Ԫ�غ���=
| 0.0728g |
| 2.0g |
�ʴ�Ϊ��3.64��
���������⿼��������ˮ���Ӧ�ã�����ʵ�����Ʊ��������������ʵķ����жϣ���Һ���ƵIJ��������ѡ�ζ�ʵ��ļ���Ӧ�ã���Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
 ��2012?����һģ���±��������ʻ�����Ĵ�����ϵ������ͼ��ʾ��ϵ���ǣ�������
��2012?����һģ���±��������ʻ�����Ĵ�����ϵ������ͼ��ʾ��ϵ���ǣ�������