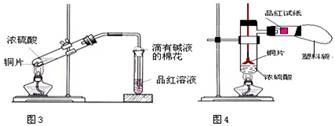
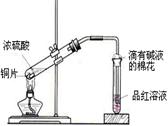
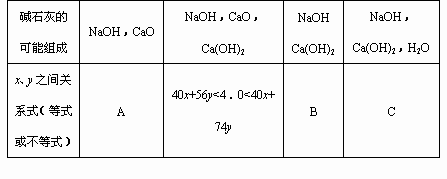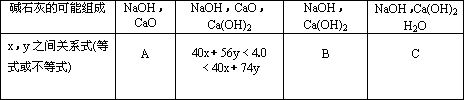��Ŀ����
��10�֣���ѧ��ѧ�̲��������½��ܣ�����������Ũ��Һ�м��������ƣ����ȣ��Ƴɵİ�ɫ������Ǽ�ʯ�ҡ��Ը�����Ŀ��Ҫ�ش��������⣺
��1�����ּ�ʯ���У���NaOH����ܺ��е������ǣ����������ɲ�����Ҳ�ɲ��䣩
���1 | ���2 | ���3 | ���4 | ���5 |
|
|
|
|
|
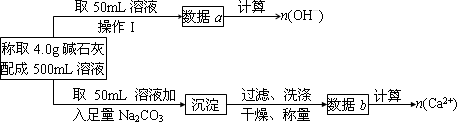
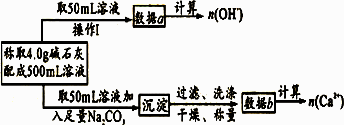
��2��Ϊ̽�����ּ�ʯ�ҵ���ɣ�ijУ�о���ѧϰС�飨�ף���Ƶ�˼·�ǣ���ȡ4.0g��ʯ�����500mL��Һ����pH�Ʋ���Һ��pH��Ȼ����ͨ�� ����ʵ�鷽������ �������������ƣ��������������ɵijɷ֡�
��3���о���ѧϰС�飨�ң�����������ϣ�����������250��ʱ���ֽ⣬����������580��ʱ���ֽ⡣������������·������ó�������ݣ�ȡ�÷��Ƶõļ�ʯ��4.0g����250��ʱ���������أ���ù�������������0.6g��ʣ�������580��ʱ�������������أ����������ּ�����0.7g ����ͨ������ȷ���ü�ʯ�Ҹ��ɷֵ�����������
��4��Ϊʹ�÷��Ƶõļ�ʯ�������Ч�����ã������ƺ�NaOH��CaO������⣬������Ƶ�ʵ��������ʲô��
��1��CaO ��Ca(OH)2��CaO��Ca(OH)2 �� Ca(OH)2��H2O��3�֣�
��2����������Ca2+�����ʵ�������2�֣�
��3��NaOH%=13%��1�֣���Ca(OH)2=72%��1�֣���H2O=15%��1�֣�
��4���ڲ��Ͻ��������³�ּ��ȣ�580�棩�����أ�2�֣�

 ��ѧ����ϵ�д�
��ѧ����ϵ�д� �ο�������ϵ�д�
�ο�������ϵ�д�
| |||||||||||