��Ŀ����
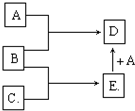 ��֪A��B��C��Ϊ�����ĵ��ʣ�����AΪ������B��CΪ�ǽ�������һ���������ת����ϵ��ͼ��ʾ����Ӧ�����Ͳ��ֲ�����ʡ�ԣ�
��֪A��B��C��Ϊ�����ĵ��ʣ�����AΪ������B��CΪ�ǽ�������һ���������ת����ϵ��ͼ��ʾ����Ӧ�����Ͳ��ֲ�����ʡ�ԣ�
��ش�
��1����������B��C��Ϊ���壬DΪ��ɫ���壬E�ĵ���ʽΪ______��A��E��һ�������·�Ӧ����D�Ļ�ѧ����ʽΪ______��
��2����������BΪ���壬CΪ��ɫ���壬�ɽ���A��ԭ�ӽṹʾ��ͼΪ______��E�ĽṹʽΪ______��A��E��һ�������·�Ӧ����D�Ļ�ѧ����ʽΪ______��
�⣺��1��B��CΪ��̬�ǽ�������Χ��С����������A����������Ӧ���ɺ�ɫ�����ֻ��O2�������ɫ�����ﳣ����ֻ��FeO��Fe3O4��CuO������ֻ��Fe3O4�������⣬��AΪFe��BΪO2��DΪFe3O4����ת��B+C��E��E+A��D��֪��CΪH2��EΪH2O������ת����ϵ��
H2O����ԭ������ԭ��֮���γ�1�Թ��õ��Ӷԣ�����ʽΪ��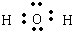 ��
��
Fe�ڸ��µ���������ˮ������Ӧ������������������������Ӧ����ʽΪ��3Fe+4H2O��g�� Fe3O4+4H2��
Fe3O4+4H2��
�ʴ�Ϊ�� ��3Fe+4H2O��g��
��3Fe+4H2O��g�� Fe3O4+4H2��
Fe3O4+4H2��
��2��������ɫ�ǽ���Ϊ̼�����룺Mg+O2��MgO��CO2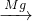 MgO��֪��AΪMg��BΪO2��CΪ̼��DΪMgO��EΪCO2������ת����ϵ��
MgO��֪��AΪMg��BΪO2��CΪ̼��DΪMgO��EΪCO2������ת����ϵ��
Mgԭ�Ӻ��������Ϊ12����3�����Ӳ㣬����������Ϊ12��ԭ�ӽṹʾ��ͼΪ��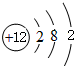 ��
��
CO2��̼ԭ������ԭ��֮���γ�2�Թ��õ��Ӷԣ��ṹʽΪ��O�TC�TO��
Mg��CO2��ȼ������̼��MgO����Ӧ��ѧ����ʽΪ��2Mg+CO2 C+2MgO��
C+2MgO��
�ʴ�Ϊ�� ��O�TC�TO��2Mg+CO2
��O�TC�TO��2Mg+CO2 C+2MgO��
C+2MgO��
��������1��B��CΪ��̬�ǽ�������Χ��С����������A����������Ӧ���ɺ�ɫ�����ֻ��O2�������ɫ�����ﳣ����ֻ��FeO��Fe3O4��CuO������ֻ��Fe3O4�������⣬��AΪFe��BΪO2��DΪFe3O4����ת��B+C��E��E+A��D��֪��CΪH2��EΪH2O������ת����ϵ��
��2��������ɫ�ǽ���Ϊ̼�����룺Mg+O2��MgO��CO2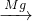 MgO��֪��AΪMg��BΪO2��CΪ̼��DΪMgO��EΪCO2������ת����ϵ��
MgO��֪��AΪMg��BΪO2��CΪ̼��DΪMgO��EΪCO2������ת����ϵ��
����������������ͼ�����ʽ����Mg��Fe��Ԫ�ص��ʼ��仯����֮����ת����ϵ���Ѷ��еȣ�ּ�ڿ���ѧ����֪ʶ���յ������̶ȣ������Ŀ��Ϣ���в²���֤���ɣ�
H2O����ԭ������ԭ��֮���γ�1�Թ��õ��Ӷԣ�����ʽΪ��
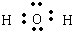 ��
��Fe�ڸ��µ���������ˮ������Ӧ������������������������Ӧ����ʽΪ��3Fe+4H2O��g��
 Fe3O4+4H2��
Fe3O4+4H2���ʴ�Ϊ��
 ��3Fe+4H2O��g��
��3Fe+4H2O��g�� Fe3O4+4H2��
Fe3O4+4H2����2��������ɫ�ǽ���Ϊ̼�����룺Mg+O2��MgO��CO2
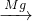 MgO��֪��AΪMg��BΪO2��CΪ̼��DΪMgO��EΪCO2������ת����ϵ��
MgO��֪��AΪMg��BΪO2��CΪ̼��DΪMgO��EΪCO2������ת����ϵ��Mgԭ�Ӻ��������Ϊ12����3�����Ӳ㣬����������Ϊ12��ԭ�ӽṹʾ��ͼΪ��
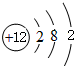 ��
��CO2��̼ԭ������ԭ��֮���γ�2�Թ��õ��Ӷԣ��ṹʽΪ��O�TC�TO��
Mg��CO2��ȼ������̼��MgO����Ӧ��ѧ����ʽΪ��2Mg+CO2
 C+2MgO��
C+2MgO���ʴ�Ϊ��
 ��O�TC�TO��2Mg+CO2
��O�TC�TO��2Mg+CO2 C+2MgO��
C+2MgO����������1��B��CΪ��̬�ǽ�������Χ��С����������A����������Ӧ���ɺ�ɫ�����ֻ��O2�������ɫ�����ﳣ����ֻ��FeO��Fe3O4��CuO������ֻ��Fe3O4�������⣬��AΪFe��BΪO2��DΪFe3O4����ת��B+C��E��E+A��D��֪��CΪH2��EΪH2O������ת����ϵ��
��2��������ɫ�ǽ���Ϊ̼�����룺Mg+O2��MgO��CO2
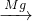 MgO��֪��AΪMg��BΪO2��CΪ̼��DΪMgO��EΪCO2������ת����ϵ��
MgO��֪��AΪMg��BΪO2��CΪ̼��DΪMgO��EΪCO2������ת����ϵ������������������ͼ�����ʽ����Mg��Fe��Ԫ�ص��ʼ��仯����֮����ת����ϵ���Ѷ��еȣ�ּ�ڿ���ѧ����֪ʶ���յ������̶ȣ������Ŀ��Ϣ���в²���֤���ɣ�

��ϰ��ϵ�д�
 Сѧ��ʱ��ѵϵ�д�
Сѧ��ʱ��ѵϵ�д�
�����Ŀ
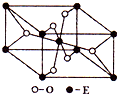 ��֪A��B��C��D��E����Ԫ�ص�ԭ���������ε�����A��B��C��Dλ��ǰ�����ڣ�Aλ�����ڱ���s������ԭ���е��Ӳ�����δ�ɶԵ�������ͬ��B�Ļ�̬ԭ���е���ռ������������ͬ��ԭ�ӹ������ÿ�ֹ���еĵ���������ͬ��Dԭ�ӵĺ���ɶԵ�������δ�ɶԵ�������3����A��B��D����Ԫ����ɵ�һ�ֻ�����M����װ�����г����е�һ���к����壬A��B����Ԫ����ɵ�ԭ�Ӹ�����Ϊ1��1�Ļ�����N�dz������л��ܼ���E�С����������֮�ƣ�E4+���Ӻ��ԭ�ӵĺ�������Ų���ͬ��
��֪A��B��C��D��E����Ԫ�ص�ԭ���������ε�����A��B��C��Dλ��ǰ�����ڣ�Aλ�����ڱ���s������ԭ���е��Ӳ�����δ�ɶԵ�������ͬ��B�Ļ�̬ԭ���е���ռ������������ͬ��ԭ�ӹ������ÿ�ֹ���еĵ���������ͬ��Dԭ�ӵĺ���ɶԵ�������δ�ɶԵ�������3����A��B��D����Ԫ����ɵ�һ�ֻ�����M����װ�����г����е�һ���к����壬A��B����Ԫ����ɵ�ԭ�Ӹ�����Ϊ1��1�Ļ�����N�dz������л��ܼ���E�С����������֮�ƣ�E4+���Ӻ��ԭ�ӵĺ�������Ų���ͬ��