��Ŀ����
��һ����ĩ���ܺ�K+��Fe3+��Al3+��Cl-��SO42-��CO32-�е������֣��ֽ�������ʵ�飺
��1��ȡ�������壬����ϡ������裬����ȫ���ܽ⣬û������ų���
��2����1����Һ�м�������Ba(OH)2��Һ��������ɫ���������˺�����Һ����HNO3�ữ���ٵ���AgNO3��Һ���а�ɫ�������ɣ�
��3��ȡ��2���е���ɫ��������������ϡ�������ȫ���ܽ⣻
��4������ȡ�������������������ˮ�������ȫ���ܽ⣬�õ�������Һ��
��5����4������Һ�м��˰�ˮʹ��Һ�ʼ��ԣ��г������ɣ����ˡ����õ��ij����м��˹�����NaOH��Һ���������١�
��������ʵ�飬�����ĩ��һ�������е�������________���϶����е�������________������ȷ���Ƿ��е�������_________����ͨ��_______����һ��ȷ�������ӡ�
 ������ϵ�д�
������ϵ�д�ij��Һ�к��д����� K+��Cl�� �� X�� Y�������ӣ��������Һ���� W�Լ�������ʵ������õ��Ĺ���X�� Y���ӵ��ƶ��ܳ�������
W�Լ� | ʵ������ | X��Y���ӵ��Ʋ� | |
A | KI��������Һ | ��Һ���� | Fe3+��SO42�� |
B | ��pH��ֽ��� | pH=7�����£� | CH3COO����Na+ |
C | �����ữ��AgNO3��Һ | ��ɫ���� | CO32����NH4+ |
D | ����NaOH��Һ | ���������� | Al3+��Mg2+ |
Ϊ��ǿ������ʴ�ԣ�����Ǧ����Ϊ��ӵ�Դ����Al��������Pb�����������ϡ���ᣬʹ�����������Ĥ�����䷴Ӧԭ�����£�
Ǧ���أ�Pb(s)+PbO2(s)+2H2SO4(aq)==2PbSO4(s)+2H2O(l)��
���أ�2Al+3H2O Al2O3+3H2��
Al2O3+3H2��
�������У����¸����ж���ȷ����( )
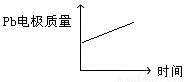
Ǧ���� | ���� | |
A | H+����Pb�缫 | H+����Pb�缫 |
B | ÿ����3molPb | ����2molAl2O3 |
C | ������PbO2+4H++2e-=Pb2++2H2O | ������2Al+3H2O��6e-=Al2O3+6H+ |
D |
|
|



 % B.
% B. % C.
% C. % D.
% D. %
%