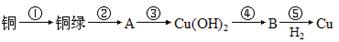
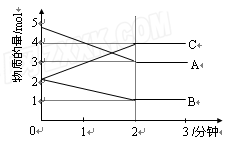
��Ŀ����
������Ũ�ȶ�Ϊ0.1 mol/L��������Һ�У����루��ͨ�룩ij���ʺ�����Ӧ�Ⱥ�˳����ȷ����
| A���ں�Fe3+��Cu2+��H+����Һ�м���п�ۣ�Cu2+��Fe3+��H+ |
| B���ں�I����SO32����Br������Һ�в���ͨ��������I����Br����SO32�� |
| C���ں�Fe3+��H+��NH4+����Һ����μ����ռ���Һ��Fe3+��NH4+��H+ |
| D���ں�AlO2����SO32����OH������Һ����μ�������������Һ��OH����AlO2����SO32�� |
D
�������������A�������ԣ�Fe3+��Cu2+��H+������������ԭ��Ӧ���Ⱥ���ɣ�������Ӧ�Ⱥ�˳��Ϊ��Fe3+��Cu2+��H+����A����B����ԭ�ԣ�SO32-��I-��Br-������������ԭ��Ӧ���Ⱥ���ɣ�������Ӧ�Ⱥ�˳��Ϊ��SO32-��I-��Br-����B����C���ȶ��ԣ�ˮ������������һˮ�ϰ����ʷ�����Ӧ�Ⱥ�˳��Ϊ��H+��Fe3+��NH4+����C����D�����ԣ�OH-��AlO2-��SO32-���ʷ�����Ӧ�Ⱥ�˳��Ϊ��OH-��AlO2-��SO32-����D��ȷ����ѡD��
���㣺���������ԡ���ԭ��ǿ���Ƚϣ����ӷ�Ӧ����������

��ϰ��ϵ�д�
�����Ŀ
���и�ѡ���е�������Ӧ,����ͬһ�����ӷ���ʽ��ʾ���� ( )
| ѡ�� | �� | �� |
| A | Ba(OH)2��Һ�����NaHCO3��Һ��� | NaOH��Һ�����NaHCO3��Һ��� |
| B | ����SO2ͨ��Ba(OH)2��Һ�� | ����SO2ͨ��Ba(OH)2��Һ�� |
| C | BaCl2��Һ��Na2SO3��Һ��� | Ba(OH)2��Һ��H2SO3��Һ��� |
| D | ������ˮ����AlCl3��Һ�� | ����AlCl3��Һ���백ˮ�� |
�������ӷ���ʽ��ѧ����ʽ��д��ȷ���� �� ��
A��ʵ�������Ȼ������ʯ���ư��� NH4++OH�� NH3��+H2O NH3��+H2O |
| B����CO2ͨ��BaCl2��Һ�� H2O +CO2+Ba2+ =BaCO3��+2H+ |
| C������SO2ͨ���Ư�۵�ˮ��Һ�� SO2+H2O+Ca2++3ClO�� = CaSO4��+2HClO+Cl�� |
D����H2��ԭMgO H2+MgO  Mg+H2O Mg+H2O |
�����£����и���������ָ����Һ��һ���ܴ����������
| A��0.1 mol��L��1 KNO3��Һ��Mg2+��Fe3+��Cl����SO42�� |
| B����ɫ������Һ��Cu2+��NH4����NO3����CO32�� |
| C��ʹpH��ֽ������Һ��K����Ca2+��Cl����ClO�� |
| D��ˮ�������c(H��)=10��12 mol��L��1����Һ��Na����NH4����SO42����NO3�� |
�����йر�����ȷ����
| A�������������ʹ��ѧƽ���ƶ� |
| B���ѷ���NO2����ƿ������ˮ�У���ɫ��dz |
| C����30%��FeCl3��Һ��ʴӡˢ��·���ϵ�ͭ����Fe3+ + Cu=Fe2+ + Cu2+ |
| D��������NaHSO4��Ba(OH)2��Һ��Ӧ��Ba2++2OH��+2H++SO42��=BaSO4��+2H2O |
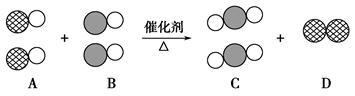
 ��ʾ̼ԭ�ӣ���
��ʾ̼ԭ�ӣ��� ��ʾ��ԭ�ӣ���
��ʾ��ԭ�ӣ��� ��ʾ��ԭ�ӣ���ͼΪ����ת�����۹��̡��������ͼʾ�ش��������⣺
��ʾ��ԭ�ӣ���ͼΪ����ת�����۹��̡��������ͼʾ�ش��������⣺