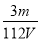��Ŀ����
��A��B��C��D������Һ���������ռ���ᡢ̼����李��Ȼ�ͭ��Һ�е�ijһ�֡���ȡ����������������ϣ�����A��B�������ɫ��״�������ɣ� C ������B��D��Ͼ�����ɫ�����������������ش��������⣺
��1��B��C�ֱ��� ��
��2����A�еμ�B�����ӷ���ʽ��
��C�еμ�D�����ӷ���ʽ��
��3������B��C��ϣ�����________�����ӷ���ʽ��_______________________________��
��4������������Ӧ������________________��Ӧ(�������Ӧ����)����Ӧ�����������IJ�ͬ���� ����ͬ���� ��
��1��NaOH NH4HCO3
��2��Cu2����2OH��=Cu(OH)2�� HCO3-��H��=CO2����H2O
��3������������ HCO3-��OH��=H2O+ CO32-
��4�����ֽ� ��һ�����ɳ������ڶ������������H2O������������H2O ��ʹ��Һ�е�����Ũ�ȼ�С
��������
��������� C ������B��D��Ͼ�����ɫ���������˵��CΪ̼����泥�A��B�������ɫ��״�������ɣ�A��B����һ��Ϊ�Ȼ�ͭ��һ��Ϊ�ռ�ۺϷ�����֪BΪ�ռNaOH����DΪ���ᡣ��A�еμ�B��Cu2����2OH��=Cu(OH)2����C�еμ�D��HCO3-��H��=CO2����H2O������NaOH��NH4HCO3���ʱHCO3-��OH��=H2O+ CO32-��������Ӧ�����ڸ��ֽⷴӦ����һ�����ɳ������ڶ������������H2O������������H2O������ʹ��Һ�е�����Ũ�ȼ�С��
���㣺���ӷ�Ӧ

 ��У����ϵ�д�
��У����ϵ�д����й����ʵķ���ش��������⣺
��1������һƿA��B�Ļ��Һ����֪���ǵ��������±���
���� | �۵�/�� | �е�/�� | �ܶ�/g��cm-3 | �ܽ��� |
A | ��11��5 | 198 | 1��11 | A��B���ܣ��Ҿ�������ˮ |
B | 17��9 | 290 | 1��26 |
�ݴ˷�������A��B�����ij��÷����ǣ� ��
��2���ڷ�Һ©������һ���л��ܼ���ȡˮ��Һ���ij����ʱ�����÷ֲ�������֪����һ��Һ������ˮ�����������һ�ּ����жϷ���: ��