��Ŀ����
�����£���20 mL 0.01 mol/L CH3COOH��Һ����μ���0.01 mol/L ��NaOH��Һ����Һ��ˮ���������c(H+)�����NaOH��Һ������仯ʾ��ͼ���£�����˵����ȷ����
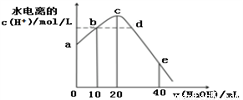
A. ��a��c���Դ���ĵ�����дٽ�����Ҳ����������
B. b��d������Һ��pH��ͬ
C. e����ʾ��Һ�У�c(Na+)=2c(CH3COO-)��2c(CH3COOH) = 0.01mol/L
D. ��b��d�Ĺ����У��ȴ���pH=7�ĵ�Ҳ����ˮ�����c(H+)=10-7�ĵ�
��ϰ��ϵ�д�
�����Ŀ
���и������ӻ�����ܴ������棬��������Ӧ�Լ�������Ӧ�����ӷ���ʽ��д��ȷ����
ѡ�� | ������ | �����Լ� | �����Լ�������Ӧ�����ӷ���ʽ |
A | NH4+��Fe2+��SO42�� | ����Ba(OH)2��Һ | 2NH4++SO42-+Ba2++2OH-��BaSO4��+2NH3��H2O |
B | Na+��OH����Cl�� | ����Mg(HCO3)2��Һ | Mg2++2HCO3-+4OH-��2H2O+Mg(OH)2��+2CO32�� |
C | K+��NH3��H2O��CO32�� | ͨ������CO2 | 2OH��+CO2��CO32��+2H2O |
D | Fe2+��NO3����HSO3�� | NaHSO4��Һ | HSO3��+H+��SO2+H2O |


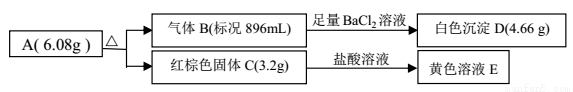
 2NH3(g) ��H��-92.0 kJ/mol������ͬ�¶��£����ܱ�������ͨ��0.5 mol N2��1.5mol H2���ﵽƽ��ʱ�ų�46.0 kJ������
2NH3(g) ��H��-92.0 kJ/mol������ͬ�¶��£����ܱ�������ͨ��0.5 mol N2��1.5mol H2���ﵽƽ��ʱ�ų�46.0 kJ������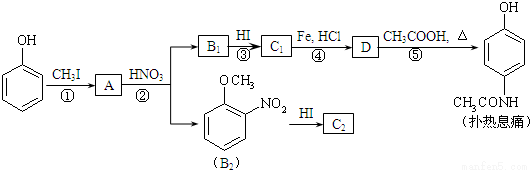
 ���밴Ҫ����գ�
���밴Ҫ����գ� ��
�� ��д����3��ͬ���칹��Ľṹ��ʽ____________��____________��____________��
��д����3��ͬ���칹��Ľṹ��ʽ____________��____________��____________��
 B.
B.  C.
C.  D.
D. 
 xC(g�� ��H��0�������������c(A)��ʱ��t�ı仯��ͼ��ʾ������˵������ȷ����
xC(g�� ��H��0�������������c(A)��ʱ��t�ı仯��ͼ��ʾ������˵������ȷ����