��Ŀ����
����Ŀ�����ڱ�ǰ�����ڵ�Ԫ��a��b��c��d��e��ԭ��������������a�ĺ����������������������ͬ��b�ļ۵��Ӳ��е�δ�ɶԵ�����3����c������������Ϊ���ڲ��������3����d��cͬ�壻e�������ֻ��1�����ӣ����������18�����ӡ��ش��������⣺
��1��b��c��d��һ�����ܴ�С��ϵΪ___________________��e����Χ�����Ų�ͼΪ_____________________��
��2��a������Ԫ���γɵĶ�Ԫ���ۻ������У����ӳ������Σ��÷��ӵ�����ԭ�ӵ��ӻ���ʽΪ_________�������мȺ��м��Թ��ۼ����ֺ��зǼ��Թ��ۼ��Ļ�������_____________���ѧʽ��д���֣���
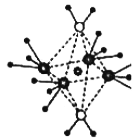
��3����5��Ԫ���γɵ�һ��1:1�����ӻ������У������ӳ�������ṹ�������ӳ����������İ�����ṹ������ͼ��ʾ�����û�������������Ϊ__________���������д��ڵĻ�ѧ��������________________��
���𰸡�N>O>S![]() sp3H2O2��N2H4SO42-���ۼ�����λ��
sp3H2O2��N2H4SO42-���ۼ�����λ��
��������
���ڱ�ǰ�����ڵ�Ԫ��a��b��c��d��e��ԭ��������������a�ĺ����������������������ͬ����a��HԪ�أ�c������������Ϊ���ڲ��������3����ԭ������������������8��Ϊ6������c��OԪ�أ�b�ļ۵��Ӳ��е�δ�ɶԵ�����3������ԭ������С��c����b��NԪ�أ�d��cͬ�壬��d��SԪ�أ�e�������ֻ��һ�����ӣ����������18�����ӣ���e��CuԪ�أ��ݴ˴��⡣
��������������a��HԪ�أ�b��NԪ�أ�c��OԪ�أ�d��SԪ�أ�e��CuԪ�ء�
(1)b��c��d�ֱ���N��O��SԪ���У�ͬһ����Ԫ���У���һ����������ԭ��������������������ƣ�����VA��Ԫ�ش�������Ԫ�أ�����N��O��S�е�һ�����ܴ�С��ϵΪN>O>S��e��CuԪ�أ�e����Χ�����Ų�ͼΪ![]() �� �ʴ�Ϊ��N>O>S��
�� �ʴ�Ϊ��N>O>S��![]() ��
��
(2)a��HԪ�أ�a������Ԫ���γɵĶ�Ԫ���ۻ������У����ӳ������Σ��÷���Ϊ���������������е�ԭ�Ӻ���3�����ۼ���һ���µ��Ӷԣ����Ը÷��ӵ�����ԭ�ӵ��ӻ���ʽΪsp3�������мȺ��м��Թ��ۼ����ֺ��зǼ��Թ��ۼ��Ļ�������H2O2��N2H4���ʴ�Ϊ��sp3��H2O2��N2H4��
(3)��5��Ԫ���γɵ�һ��1��1�����ӻ������У������ӳ�������ṹ��˵�������ӵ�����ԭ�Ӽ۲���ӶԸ�����4�Ҳ����µ��Ӷԣ�����������Ϊ��������ӣ������ӳ����������İ�����ṹ(��ͼ)������ͼ��֪�������ӵ���λ����6�����а����Ӹ�����4��ˮ���Ӹ�����2��ˮ���ӡ���������ͭ����֮������λ����ˮ���ӺͰ������ڲ��й��ۼ��������������к��й��ۼ�����λ�����ʴ�Ϊ��SO42-�����ۼ�����λ����

����Ŀ���������������������������ܹ��������·�Ӧ��
MnO![]() ��H2C2O4��H���D��Mn2����CO2����H2O(δ��ƽ)
��H2C2O4��H���D��Mn2����CO2����H2O(δ��ƽ)
��4 mL 0.001 mol��L��1KMnO4��Һ��2 mL 0.01 mol��L��1 H2C2O4��Һ���о���ͬ�����Ի�ѧ��Ӧ���ʵ�Ӱ�졣�ı�����������ʾ��
��� | 10%�������/mL | �¶�/�� | �������� |
�� | 2 | 20 | |
�� | 2 | 20 | 10�α���MnSO4��Һ |
�� | 2 | 30 | |
�� | 1 | 20 | 1 mL����ˮ |
��1���÷�Ӧ���������ͻ�ԭ�������ʵ���֮��Ϊ______________________��
��2������о������Ի�ѧ��Ӧ���ʵ�Ӱ�죬ʹ��ʵ��____________��________(������ʾ����ͬ)������о��¶ȶԻ�ѧ��Ӧ���ʵ�Ӱ�죬ʹ��ʵ��________��________��
��3���Ա�ʵ���������������о�____________�Ի�ѧ��Ӧ���ʵ�Ӱ�죬ʵ�����м���1 mL����ˮ��Ŀ����______________________________________________________________��
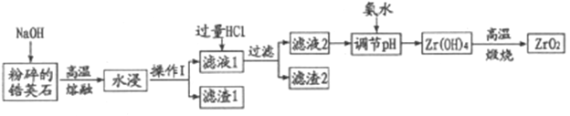
����Ŀ���������Ͼ��и�Ӳ�ȡ���ǿ�ȡ������ԡ����ߵ���ĥ�Լ��ͻ�ѧ��ʴ�Ե��������ﻯ���ܡ����Ӣʯ(��Ҫ�ɷ�Ϊ ZrSiO4����������Al2O3��SiO2��Fe2O3������)Ϊԭ��ͨ�����۷��Ʊ������(ZrO2)���������£�
25��ʱ���й�������ˮ��Һ�г���ʱ��pH���ݣ�
Fe(OH)3 | Zr(OH)4 | Al(OH)3 | |
��ʼ����ʱpH | 1.9 | 2.2 | 3.4 |
������ȫʱpH | 3.2 | 3.2 | 4.7 |
��ش��������⣺
(1)������ּ�����ѧ��Ӧ���ʵĴ�ʩ��___________��
(2)����I��������___________����2�ijɷ�Ϊ___________��
(3)�Ӣʯ��������������ת��ΪNa2ZrO3��д���÷�Ӧ�Ļ�ѧ����ʽ��___________��
(4)������pH��ʱ�����ʵ�pH��Χ��___________��Ϊ�˵õ�����ZrO2��Zr(OH)4��Ҫϴ�ӣ�����Zr(OH)4�Ƿ�ϴ�Ӹɾ��ķ�����___________��
(5)д�����������������̵Ļ�ѧ����ʽ___________������ZrO2�����ʣ��Ʋ���������;___________��