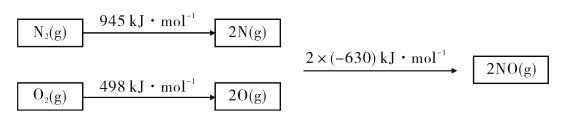
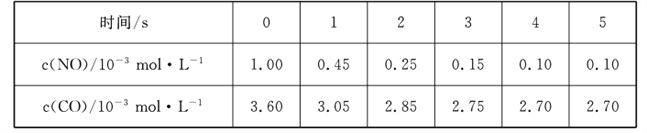
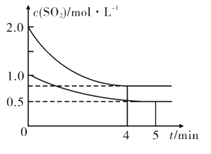
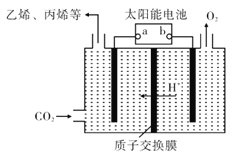
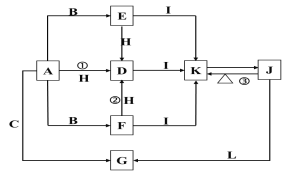
��Ŀ����
����Ŀ����NAΪ�����ӵ�������ֵ�������й�������ȷ������ ��
A. ��״���£�5.6L���������(�����������Ļ����)������Ϊ2.4g�����к���NA������
B. 5.6gFe��1L0.2mol/LHNO3��Һ��ַ�Ӧ������ʧȥԼ1.204��1023������
C. 72gCaO2��KHS�Ļ�����к��е������ӵ���ĿΪNA
D. 1molCH4��1molCl2��ַ�Ӧ������һ�ȼ���ķ�����ΪNA
���𰸡�C
��������������A����ݱ�״������������������������������He��O2���ʵ�����1��He�к�2�����ӣ�1��O2�к�16�����ӣ�B�����Fe��HNO3���ʵ���֮��ȷ����Ӧ������ȫ��Ӧ�����ʼ�����C�CaO2��KHS��Ħ����������72g/mol��CaO2��Ca2+��O22-������Ϊ1:1��KHS��K+��HS-������Ϊ1:1��D�CH4��Cl2�����·���ȡ����Ӧ����CH3Cl��CH2Cl2��CHCl3��CCl4��HCl�Ļ���
�����A�n��He��+n��O2��=![]() =0.25mol��4g/moln��He��+32g/moln��O2��=2.4g�����n��He��=0.2mol��n��O2��=0.05mol��1��He�к�2�����ӣ�1��O2�к�16�����ӣ���״����5.6L����Ϊ2.4g��������������к��������ʵ���Ϊ0.2mol
=0.25mol��4g/moln��He��+32g/moln��O2��=2.4g�����n��He��=0.2mol��n��O2��=0.05mol��1��He�к�2�����ӣ�1��O2�к�16�����ӣ���״����5.6L����Ϊ2.4g��������������к��������ʵ���Ϊ0.2mol![]() 2+0.05mol
2+0.05mol![]() 16=1.2mol��A�����B�Fe��ϡHNO3���ܷ����ķ�ӦΪFe+4HNO3��ϡ��=Fe��NO3��3+NO��+2H2O���٣���3Fe+8HNO3��ϡ��=3Fe��NO3��2+2NO��+4H2O���ڣ���n��Fe��=
16=1.2mol��A�����B�Fe��ϡHNO3���ܷ����ķ�ӦΪFe+4HNO3��ϡ��=Fe��NO3��3+NO��+2H2O���٣���3Fe+8HNO3��ϡ��=3Fe��NO3��2+2NO��+4H2O���ڣ���n��Fe��=![]() =0.1mol��n��HNO3��=1L
=0.1mol��n��HNO3��=1L![]() 0.2mol/L=0.2mol��
0.2mol/L=0.2mol��![]() =
=![]() =
=![]() ��Fe��ϡHNO3������Ӧ���ҷ�Ӧ��Fe���������ݷ�Ӧ��ÿ����8molHNO3ת��6mol���ӣ���Ӧ��ת�Ƶ������ʵ���Ϊ
��Fe��ϡHNO3������Ӧ���ҷ�Ӧ��Fe���������ݷ�Ӧ��ÿ����8molHNO3ת��6mol���ӣ���Ӧ��ת�Ƶ������ʵ���Ϊ![]() =0.15mol��ת�Ƶ�����Ϊ9.03
=0.15mol��ת�Ƶ�����Ϊ9.03![]() 1022��B�����C�CaO2��KHS��Ħ��������Ϊ72g/mol��CaO2��KHS������Ȼ�ϣ�72g����������ʵ�����Ϊ72g
1022��B�����C�CaO2��KHS��Ħ��������Ϊ72g/mol��CaO2��KHS������Ȼ�ϣ�72g����������ʵ�����Ϊ72g![]() 72g/mol=1mol��CaO2��KHS�����������Ӹ����ȶ�Ϊ1:1��72gCaO2��KHS�Ļ�����к����������ʵ���Ϊ1mol��C����ȷ��D�CH4��Cl2�����·���ȡ����Ӧ����CH3Cl��CH2Cl2��CHCl3��CCl4��HCl�Ļ������1molCH4��1molCl2��ַ�Ӧ���ɵ�CH3Cl���ʵ���С��1mol��D�����ѡC��
72g/mol=1mol��CaO2��KHS�����������Ӹ����ȶ�Ϊ1:1��72gCaO2��KHS�Ļ�����к����������ʵ���Ϊ1mol��C����ȷ��D�CH4��Cl2�����·���ȡ����Ӧ����CH3Cl��CH2Cl2��CHCl3��CCl4��HCl�Ļ������1molCH4��1molCl2��ַ�Ӧ���ɵ�CH3Cl���ʵ���С��1mol��D�����ѡC��