��Ŀ����
������������ճ������м�Ϊ�������ᣬ��һ��������CH3COOH��Һ�д��ڵ���ƽ�⣺CH3COOH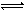 CH3COO��+H+ ��H��0
CH3COO��+H+ ��H��0
��1�������£�pH��5������Һ�У�c(CH3COO��)��______mol/L(��ȷֵ��Ҫ����ʽ���ػ���)��
��2�����з����п���ʹ0.10 mol��L-1 CH3COOH�ĵ���̶��������
a����������0.10 mol��L��1ϡ���� b������CH3COOH��Һ c����ˮϡ����0.010 mol��L��1
d���������������� e�����������Ȼ��ƹ��� f����������0.10 mol��L��1 NaOH��Һ
��3������������пͶ��������pH������3�Ĵ����������Һ�У�������ַ�Ӧ����ֻ��һ����Һ����п��ʣ�࣬�����������������V(����)_________V(����)������ڡ�����С�ڡ����ڡ���
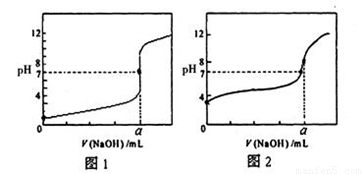
��4����NaOH��Һ�ֱ�ζ�20.00mL0.1mol/L�����20.00mL0.1mol/L������Һ���õ���ͼ��ʾ�����ζ����ߣ���NaOH��Һ�ζ�������Һ�������� ���ͼ1����ͼ2����
��5�������£���0.1 mol/L�����0.1 mol/L��������Һ��ϣ�������ҺΪ���ԣ�������Һ�и����ӵ�Ũ�Ȱ��ɴ�С����Ϊ_______________________________��
��6����ͼ��ʾ��Һ��c(H��)��c(OH��)�Ĺ�ϵ
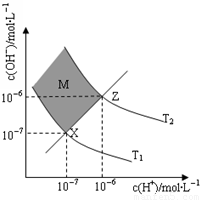
��M�����ڣ���Ӱ���֣������c(H��)______c(OH��)������ڡ�����С�ڡ����ڡ���
����T2�¶��£���pH��9 NaOH��Һ��pH��4 HCl��Һ��ϣ������û����Һ��pH��7����NaOH��Һ��HCl��Һ�������Ϊ ������Ϻ���Һ����ı仯���Բ��ƣ�
��1��10��5��10��9��2��bcf��3��С�ڣ�4��ͼ2
��5��c(Na+)��c(CH3COO��)��c(Cl��)��c(H��)��c(OH��)��6������ ����1��9
��������
�����������1���� pH=5��ϡ������Һ�У�c��H+��=10-5mol/L��c��OH-��=10-9mol/L��������Һ����غ��֪c��H+��=c��CH3COO-��+c��OH-������c��CH3COO-��=��10-5-10-9��mol/L��2�����ȡ���ˮϡ�͡�����������ʾ���ʹ����ƽ�������ƶ����ʴ�Ϊ bcf����3�����������ᣬ�������ͽ����ķ�Ӧ������ƽ�ⲻ�ϵ������ƶ�������������������ӣ����Դ����������������ϴʴ�ΪС�ڣ���4���ζ���ʼʱ0.002mol/L����pH=3��0.002mol/L����pH��3�����Եζ������������ͼ2����5����0.1mol/L�����0.1mol/L��������Һ��ϣ���Ӧ���ɴ��ᣬ������������Һ�����ԣ���Һ�����ԣ�Ӧ�����������ᣬ���ɴ��ᣬ��c��Na+����c��CH3COO-����c��Cl-����Һǡ�ó����ԣ������Ӻ���������Ũ��һ����ȣ�c��H+��=c��OH-��������c��Na+����c��CH3COO-����c��Cl-����c��H+��=c��OH-������6������ͼ��֪��ZX�߱�ʾc(H��)=c(OH��)����ZX���ϲ��ֱ�ʾc(H��)<c(OH��)������ͼ֪����T2�¶��£�ˮ�����ӻ�Ϊ��c��H+��•c��OH-��=10-12�����¶���pH=9��NaOH��Һ��c��OH-��=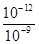 =10-3mol/L��pH=4��HCl��Һc��H+��=10-4mol/L�������û����Һ��pH=7����Ӧ�����Һ��c��OH-��=
=10-3mol/L��pH=4��HCl��Һc��H+��=10-4mol/L�������û����Һ��pH=7����Ӧ�����Һ��c��OH-��=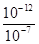 =10-5mol/L����
=10-5mol/L����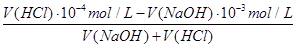 =10-5
mol/L����V(NaOH)��V(HCl)=1��9��
=10-5
mol/L����V(NaOH)��V(HCl)=1��9��
���㣺���������ˮ�еĵ���ƽ�⣻����Ũ�ȴ�С�ıȽϣ��к͵ζ������ӻ�����

 CH3COO��+H+ ��H��0��
CH3COO��+H+ ��H��0�� CH3COO��+H+
��H��0��
CH3COO��+H+
��H��0�� CH3COO��+H+ ��H��0��
CH3COO��+H+ ��H��0��