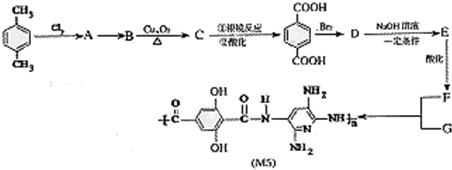
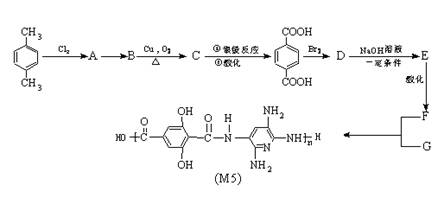
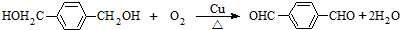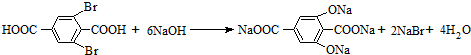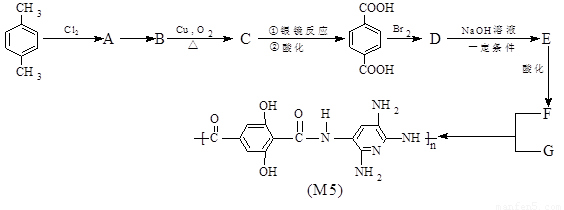��Ŀ����
M5��ά�����������ĨD�ֳ���������ά������ͬ�ķ��������£�M5��ά�����еķ���Ƭ������35�����ҷ��Ⱥͷ�������ʮ�ֳ�ɫ���ݱ���������ʿ�������ͷ��.�������ĺ���ǰ���������ս������ס����������ʿ����������M5��ά�Ǹո�������ǿ�����Ӽ�����ľۺ����z�Ǿۺ�����������ʱ����x����y��������侧��ṹ��������������M5��ά�ĺϳ�·�ߣ�(��Щ��Ӧδע����ʲ)
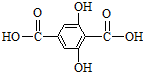
 ��˼����(1)�ϳ�M5�ĵ���Ľṹ��ʽF ______________��G ______________��
��˼����(1)�ϳ�M5�ĵ���Ľṹ��ʽF ______________��G ______________��
(2)��Ӧ���ͣ�A��B______________��B��C______________��
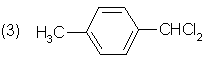
(3)Aͬʱ�������ɵ�ͬ���칹��______________ ��
(4)д����ѧ��Ӧ����ʽ��
C���Ա������__________________________________________��
D��E��____________________________ ��
(5)1mol��F�� NaHCO3��Һ��Ӧ������NaHCO3______________ mol��
(6)��������M5��ά���Ӽ���ǿ�����?
��

��2��ȡ����ˮ�⣩��������Ӧ

��5��2mol
(6)-OH��-NH2��֮�������������γ������