��Ŀ����
��Ԫ��������������ʼ�ת���ij���Ԫ�ء�
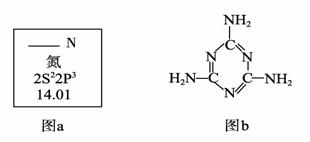
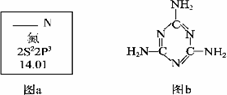
��1��ͼa��Ԫ�����ڱ���NԪ�ص��й���Ϣ��ͼa�к�����ȱʧ�ľ����������������� ��
15N��NԪ�ص�һ����Ҫ���أ���һ��ԭ�Ӻ��к��е�������Ϊ�������� ��
��2�������谷�Ľṹ��ʽ����ͼb����������˽⣬���й��������谷�ı����У�
��ȷ������������ (����ĸ���)��
A�������谷���۵���ܸܺߡ� B�������谷�ĺ������ߴ�67%����
������ C�������谷����������� D�������谷����������ԭ����ͬһƽ����
��3���������ѧ���Ǻϳ����ɵ�Ԫ���γɵ�N5n+������ʽ![]() Ϊ
Ϊ
��nֵΪ������ ��
��H��N���γɻ�����NH5 ����֪��������ˮ��Ӧ��H2���ɣ���NH5�к��еĻ�ѧ��Ϊ���������������� ��
��4����֪MΪ��Ԫ����һ�ֽ���Ԫ����ɵ����ӻ�������н���Ԫ�ص���������Ϊ
35��4����M��������ȫ����������Ҫ�����á�������GΪ����ɫ���塣HΪһ�ֳ�����Һ�壬A��B��C��XΪ���ʣ�����A��C��XΪ���壬A��X��Ϊ�����гɷ֡�I��JΪ�����Ĺ�ҵԭ�ϡ�
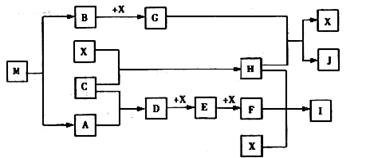
��д��D��X��Ӧ�Ļ�ѧ����ʽ����������������������������������
��д��G��H��Ӧ�����ӷ���ʽ������������������������������������ ��
��ʵ������IӦ��α��棿�� ������������������������������ ��
�ܳ�����M��ײ��ʱ�ɷֽ⣬13gM��ȫ�ֽ�ΪA��Bʱ���ų�akJ��������д��M�ֽ���Ȼ�ѧ����ʽ�������������������������������������������� ��
��M��һ��������ˮ���Σ���ˮ��Һ�������ԣ������ӷ���ʽ����ԭ������������������ ��
(1) 7 ��8�� (2) B
��3����1 �� ���Ӽ��ͼ��Լ������ۼ�����������ÿ��1�֣�
��4��
��4NH3+5O2 ![]() 4NO+6H20���������� ��2�֣�
4NO+6H20���������� ��2�֣�
��2Na2O2 + 2H2O === 4NaOH + O2���� ��2�֣�
����ɫ�Լ�ƿ�������䰵������ ��1�֣�
��2NaN3(s) = 2Na(s) + 3N2(g)����H = 10akJ/mol�� ��2�֣�
��N3 + H2O��![]() �� HN3 + OH �������������������� ��1�֣�
�� HN3 + OH �������������������� ��1�֣�

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
| |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||




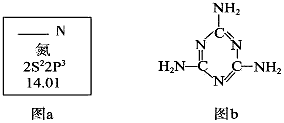
 ��nֵΪ_________��
��nֵΪ_________��
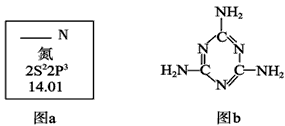
 ����nֵΪ_______��
����nֵΪ_______��