��Ŀ����
���й�������һ�����ʵ���Ũ�ȵ���Һ˵����ȷ���ǣ� ��
A. ��NaCl��������������ƽ���̳������������루8g�������루0��6g�������ĺ�Ϊ8��6g
B. ����ƿ�ϱ��п̶��ߡ��¶Ⱥ�Ũ�ȣ�ʹ��ǰҪ���ȼ���Ƿ�©ˮ
C. ����һ�����ʵ���Ũ�ȵ���Һ�õ�����Ҫ�����У�������ƽ����Ͳ���ձ���������������ƿ����ͷ�ιܡ�
D. ����1L0.2mol/L��H2SO4 ��Һ������ʱ���ӿ̶��߲����������Ũ��ƫ��
��ϰ��ϵ�д�
�����Ŀ





 ����Cl��
����Cl�� ��
�� ��SCN��
��SCN��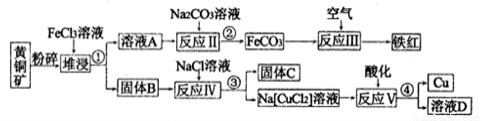


 һ2
һ2 ===
===