��Ŀ����
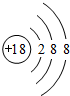
9����֪X��Y��Z��W�Ƕ�����Ԫ���е����ַǽ���Ԫ�أ����ǵ�ԭ��������������XԪ�ص�ԭ���γɵ������Ӿ���һ�����ӣ�Z��W��Ԫ�����ڱ��д������ڵ�λ�ã����ǵĵ����ڳ����¾�Ϊ��ɫ���壬Yԭ�ӵ��������������ڲ��������2������1����д��Z��Ԫ�����ڱ��е�λ�ã��ڶ����ڵڢ�A�壮
��2��һ�������£���X������Z���ʷ�Ӧ����1mol E�ų�������Ϊ46.2kJ��д��������Eͨ�뵽��������Һ�е����ӷ���ʽAl3++3NH3•H2O=Al��OH��3��+3NH4+��
��3����X��Y��Z��W����Ԫ�ؿ������ʽ�Σ��û������ˮ��Һ������NaOH��Һ�ڼ��������·�Ӧ�����ӷ���ʽΪNH4++HCO3-+2OH-$\frac{\underline{\;\;��\;\;}}{\;}$NH3��+2H2O+CO32-��
��4����ҵ����E�������������Ĺܵ��Ƿ�©�����ɹ۲쵽�������̣�ͬʱ��Z�ĵ������ɣ�д����Ӧ�Ļ�ѧ����ʽ��8NH3+3Cl2=6NH4Cl+N2���÷�Ӧ�б�������E����뷴Ӧ��E������֮��Ϊ1��4��
���� X��Y��Z��W�Ƕ����������ַǽ���Ԫ�أ����ǵ�ԭ��������������XԪ��ԭ���γɵ����Ӿ���һ�����ӣ���X��HԪ�أ�Yԭ�ӵ��������������ڲ��������2�����������Ӳ��ܳ���8��ֻ����2�����Ӳ㣬����������Ϊ4����Y��CԪ�أ�Z��W��Ԫ�����ڱ��д������ڵ�λ�ã����ǵĵ����ڳ����¾�Ϊ��ɫ���壬��Z��ԭ������С��W������Z��NԪ�أ�W��OԪ�أ��ݴ˽��
��� �⣺X��Y��Z��W�Ƕ����������ַǽ���Ԫ�أ����ǵ�ԭ��������������XԪ��ԭ���γɵ����Ӿ���һ�����ӣ���X��HԪ�أ�Yԭ�ӵ��������������ڲ��������2�����������Ӳ��ܳ���8��ֻ����2�����Ӳ㣬����������Ϊ4����Y��CԪ�أ�Z��W��Ԫ�����ڱ��д������ڵ�λ�ã����ǵĵ����ڳ����¾�Ϊ��ɫ���壬��Z��ԭ������С��W������Z��NԪ�أ�W��OԪ�أ�
��1��ZΪ��Ԫ�أ�ԭ�Ӻ�����2�����Ӳ㣬�����5�����ӣ����ڵڶ����ڵڢ�A�壬�ʴ�Ϊ���ڶ����ڵڢ�A�壻
��2��һ�������£���X������Z���ʷ�Ӧ���� EΪNH3��������NH3ͨ�뵽��������Һ�е����ӷ���ʽ��Al3++3NH3•H2O=Al��OH��3��+3NH4+���ʴ�Ϊ��Al3++3NH3•H2O=Al��OH��3��+3NH4+��
��3��H��C��N��O����Ԫ�ؿ������ʽ��NH4HCO3��̼�����������ŨNaOH��Һ��Ӧ����̼���ơ�������ˮ����Ӧ���ӷ���ʽΪ��NH4++HCO3-+2OH-$\frac{\underline{\;\;��\;\;}}{\;}$NH3��+2H2O+CO32-���ʴ�Ϊ��NH4++HCO3-+2OH-$\frac{\underline{\;\;��\;\;}}{\;}$NH3��+2H2O+CO32-��
��4����ҵ����E�������������Ĺܵ��Ƿ�©�����ɹ۲쵽�������̣�ͬʱ�е��ʵ������ɣ���ѧ����ʽΪ��8NH3+3Cl2=6NH4Cl+N2������Ӧ����8mol�����μӷ�Ӧ��ֻ��2mol���������÷�Ӧ�б�������NH3����뷴Ӧ��NH3������֮��Ϊ2mol��8mol=1��4���ʴ�Ϊ��8NH3+3Cl2=6NH4Cl+N2��1��4��
���� ���⿼��λ�ýṹ���ʵĹ�ϵ��Ӧ�ã�Ԫ�ص��ƶ�Ϊ���Ĺؼ������ضԻ�ѧ����Ŀ��飬�Ѷ��еȣ�

| A�� | Cl- | B�� | Br- | C�� | I- | D�� | Cl2 |
| A�� | ������������ͭ��Һ�У���Һ����ɫ��Ϊdz��ɫ | |
| B�� | ��������ȼ�ŵ�����������ƿ�У������ɻ�ɫ��Ϊ����ɫ | |
| C�� | ���¼���ľ̿������ͭ�Ļ��������ɺ�ɫ��ɺ�ɫ | |
| D�� | ��ˮ�м�������������أ�Һ������ɫ����Ϻ�ɫ |
���� ���� | ��A | ��A | ��A | ��A | ��A | ��A | ��A | 0 |
| 2 | �� | �� | �� | |||||
| 3 | �� | �� | �� | �� | �� | |||
| 4 | �� | �� |
��2������������ˮ���������ǿ�����ʵĵ���ʽ
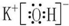 ����ѧ�������Ӽ������ۼ�
����ѧ�������Ӽ������ۼ���3���õ���ʽ��ʾ��Ԫ�����Ԫ���γɻ�����Ĺ���

��4���١��ڡ��ޡ�������Ԫ������������ˮ����������ǿ����HClO4���ѧʽ��
��5����Ԫ�����Ԫ�����ߺ˵����֮����26��
| X | ��ֲ����������ȱ�ٵ�Ԫ�أ�����ɵ����ʵ���ҪԪ�� |
| Y | �ؿ��к����ӵ�һλ |
| Z | ����������ԭ�Ӱ뾶��� |
| Q | �����д���ʹ����Ͻ���Ʒ����ҵ�Ͽ��õ����������ķ����Ʊ� |
| M | ��ˮ�д���������Ԫ��֮һ������������ϼ��븺�۵Ĵ�����Ϊ6 |
 ��
����2����֪37Rb��53I��λ�ڵ������ڣ��ֱ���Z��Mͬһ���壮�����й�˵����ȷ����ACD������ţ���
A��ԭ�Ӱ뾶��Rb��I
B��RbM�к��й��ۼ�
C����̬�⻯�����ȶ��ԣ�M��I
D��Rb��Q��M������������Ӧ��ˮ�����������������Ӧ
��3����XM3���ˮ�������XH3��HMO����X��MԪ�صĵ縺�Դ�С˳���ǣ�X��M�������������=��������֪����MԪ�ص縺�Թ�ϵ�ǣ�P��M����PM3ˮ��Ļ�ѧ��Ӧ����ʽ��PCl3+3H2O=3HCl+H3PO3��
��4��X��Y��ɵ�һ����ɫ������������Ϊ����ɫ������״����40L����ɫ������15L����ͨ��һ��Ũ�ȵ�NaOH��Һ�У�ǡ�ñ���ȫ���գ�ͬʱ���������Σ���д���÷�Ӧ�����ӷ���ʽ8NO+3O2+8OH-=2NO3-+6NO2-+4H2O��
| A�� | ���Ӻͱ��״� | B�� | �Ҵ����Ҷ��� | C�� | C4H8��C5H10 | D�� | ������Ӳ֬�� |
| ���� | C | H2 | CO |
| ��H/kJ•mol-1 | -393.5 | -285.8 | -283.0 |
��д��C��ȫȼ�յ��Ȼ�ѧ����ʽC��s��+O2��g��=CO2��g����H=-393.5 kJ/mol��
����ͬ�����£���ͬ�����������һ����̼��ȫȼ�գ��ų������϶����H2��
��д��úת��Ϊˮú������Ҫ��Ӧ���Ȼ�ѧ����ʽ��C��s��+H2O��g��=CO��g��+H2��g����H=+87.5kJ/mol��
| A�� | CS2 | B�� | H2O | C�� | CH3CH2Cl | D�� | NH3 |