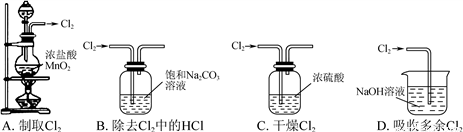
��Ŀ����
��80��ʱ����0.4mol�������������������2L�ѳ�յĹ̶��ݻ����ܱ������У���һ��ʱ��Ը������ڵ����ʽ��з������õ��������ݣ�
ʱ�䣨s�� C��mol/L�� | 0 | 20 | 40 | 60 | 80 | 100 |
C��N2O4�� | 0. 20 | a | 0.10 | c | d | e |
C��NO2�� | 0.00 | 0.12 | b | 0.22 | 0.22 | 0.22 |
��Ӧ������100s��Ӧ�������¶Ƚ��ͣ������������ɫ��dz��
��1���÷�Ӧ�Ļ�ѧ����ʽΪ__________________________������b_________c���<������=������>������
��2��20sʱ��N2O4�ĵ�Ũ��Ϊ__________________mol��L��1��0��20s��N2O4��ƽ����Ӧ����Ϊ________________��
��3���÷�Ӧ��ƽ�ⳣ������ʽK=___________________
��80��ʱ�÷�Ӧ��ƽ�ⳣ��KֵΪ:______________��������С�����2λ����
��4��������������ͬʱ���÷�Ӧ��KֵԽ��������ƽ��ʱ____________��
A.N2O4��ת����Խ�� B.NO2�IJ���Խ��
C.N2O4��NO2��Ũ��֮��Խ�� D.����Ӧ���еij̶�Խ��
 �Ǽ�����������ϵ�д�
�Ǽ�����������ϵ�д���ҵ����ú��ˮΪԭ��ͨ��һϵ��ת���ɱ�Ϊ�����Դ������ҵԭ�ϼ״���
��1����֪��C(s)+O2(g)=CO2(g) ��H1
��2H2(g)+O2(g)=2H2O (l) ��H2
��H2O (l)= H2O (g) ��H3
��̼��ˮ������ӦC(s)+2H2O(g) CO2(g)+2H2(g)�Ħ�H =________��
CO2(g)+2H2(g)�Ħ�H =________��
��2����ҵ��Ҳ���Խ�����������Ӧ�õ���CO2��H2��һ���ϳɼ״�����Ӧ����ʽΪ��CO2(g)��3H2(g) CH3OH(g)��H2O(g����H��0
CH3OH(g)��H2O(g����H��0
�ٹ�ҵ����������CO2��H2��ת����________���ǰ�ߴ������ߴ���һ�������жϡ�����Ϊ����״��IJ��ʿ��Բ�ȡ�Ĵ�ʩ��_______________�������㣩��
����һ���º����ܱ������г���1 mol CO2��3 mol H2����������Ӧ�����CO2��CH3OH(g)Ũ����ʱ��仯����ͼ��ʾ�����¶��µ�ƽ�ⳣ��Ϊ______��������λ��Ч���֣���
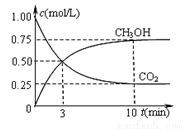
�ı��¶ȣ�ʹ��ӦCO2(g)+3H2(g) CH3OH(g)+H2O(g)�е��������ʶ�Ϊ��̬����ʼ�¶������ͬ��T1�桢2 L�ܱ�����������Ӧ�����в������ݼ��±���
CH3OH(g)+H2O(g)�е��������ʶ�Ϊ��̬����ʼ�¶������ͬ��T1�桢2 L�ܱ�����������Ӧ�����в������ݼ��±���
��Ӧʱ�� | CO2��mol�� | H2��mol�� | CH3OH��mol�� | H2O��mol�� | |
��Ӧ�� ���º��� | 0min | 2 | 6 | 0 | 0 |
10min | 4.5 | ||||
20min | 1 | ||||
30min | 1 | ||||
��Ӧ�� ���Ⱥ��� | 0min | 0 | 0 | 2 | 2 |
�ٴﵽƽ��ʱ����Ӧ��Աȣ�ƽ�ⳣ��K(��)___K(��)�����������������=������ͬ����ƽ��ʱCH3OH��Ũ��c(��)___c(��)��
�ڶԷ�Ӧ��ǰ10 min�ڵ�ƽ����Ӧ���ʦ�(CH3OH)=______����30 minʱֻ���������ٳ���1 mol CO2(g)��1 mol H2O(g)����ƽ��_____�ƶ��������������������