��Ŀ����
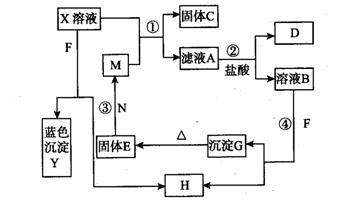
��8�֣���֪XΪ��ѧ��ѧ��һ�ֳ������Σ�FΪ����ɫ���壻M��NΪ�����Ľ�����N������������ͻ���ϣ��������������ͻ���������������������HΪ���嵥�ʣ�DΪ��ɫ���壬D�ڿ����л���ֺ���ɫ�������ʵ�ת����ϵ����ͼ(���ַ�Ӧ������ȥ)��
��ش��������⣺��1��E�Ļ�ѧʽΪ ��
��2) ��M˿����ʢ��X��Һ���Թ��У���Ӧһ��ʱ���������� ��
(3) ��Ӧ�ڵ����ӷ���ʽΪ ��
��4���ڷ�Ӧ�٢ڢۢ��������û���Ӧ���� ��
��1��Fe2O3 ��2����˿�ϸ��к�ɫ���ʣ���Һ��ɫ��dz
��3��3Fe2++NO3- +4H+ = 3Fe3+ + NO�� + 2H2O��4���٢�
����

��ϰ��ϵ�д�
�����Ŀ
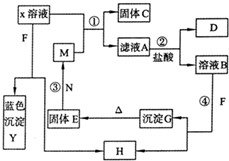 ��2009?����ģ�⣩��֪XΪ��ѧ��ѧ��һ�ֳ������Σ�FΪ����ɫ���壻M��NΪ�����Ľ�����N������������ͻ���ϣ��������������ͻ���������������������HΪ���嵥�ʣ�DΪ��ɫ���壬D�ڿ����л���ֺ���ɫ�������ʵ�ת����ϵ��ͼ�����ַ�Ӧ��������ȥ����
��2009?����ģ�⣩��֪XΪ��ѧ��ѧ��һ�ֳ������Σ�FΪ����ɫ���壻M��NΪ�����Ľ�����N������������ͻ���ϣ��������������ͻ���������������������HΪ���嵥�ʣ�DΪ��ɫ���壬D�ڿ����л���ֺ���ɫ�������ʵ�ת����ϵ��ͼ�����ַ�Ӧ��������ȥ���� ��֪XΪ��ѧ��ѧ��һ�ֳ������Σ�FΪ����ɫ���壻M��NΪ�����Ľ�����N������������ͻ���ϣ��������������ͻ���������������������HΪ���嵥�ʣ�DΪ��ɫ���壬D�ڿ����л���ֺ���ɫ�������ʵ�ת����ϵ��ͼ�������ַ�Ӧ��������ȥ����
��֪XΪ��ѧ��ѧ��һ�ֳ������Σ�FΪ����ɫ���壻M��NΪ�����Ľ�����N������������ͻ���ϣ��������������ͻ���������������������HΪ���嵥�ʣ�DΪ��ɫ���壬D�ڿ����л���ֺ���ɫ�������ʵ�ת����ϵ��ͼ�������ַ�Ӧ��������ȥ����