��Ŀ����
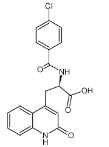
����Ŀ��ijѧϰС��������ͼװ�òⶨ��þ�Ͻ������������������������ԭ��������
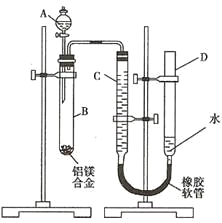
(1)A���Լ�Ϊ_______________________��
(2)ʵ��ǰ���Ƚ���þ�Ͻ���ϡ�����н���Ƭ�̣���Ŀ����_____________________��
(3)��������ԣ���ҩƷ��ˮװ��������У����Ӻ�װ�ú�����еIJ������У�
�ټ�¼C��Һ��λ�ã�
�ڽ�B��ʣ�������ˡ�ϴ�ӡ�������أ�
�۴�B�в���������������ָ������º�¼C��Һ��λ�ã�
����A��B�еμ������Լ���
�ݼ�������ԡ�����������˳����______________(�����)����¼C��Һ��λ��ʱ��������ƽ���⣬��Ӧ____________________��
(4)B�з�����Ӧ�Ļ�ѧ����ʽΪ_________________________________________��
(5)��ʵ������þ�Ͻ������Ϊa g ������������Ϊb mL(�ѻ���Ϊ��״��)��B��ʣ���������Ϊc g�����������ԭ������Ϊ______________��
(6)ʵ������У���δϴ�ӹ������õIJ����������������������____________������ƫ��������ƫС����������Ӱ��������
���𰸡�NaOH��Һ ��ȥþ�����Ͻ���������Ĥ �ݢ٢ܢۢ� ʹD��C������Һ����ƽ 2Al+2NaOH+2H2O=2NaAlO2+3H2�� ![]() ƫС
ƫС
��������
(1)Al��NaOH��Һ��Ӧ������������Mg������NaOH��Һ��Ӧ��A��װ��NaOH��Һ��
(2)Mg��Al�DZȽϻ��õĽ�����������е�������Ӧ��������þ����������Ĥ��
(3)�����������̼�ԭ���ǣ���������ԣ���¼��ʼҺ�棻����Ӧ�������������¼C�е�Һ�棬���������Ϊ����H2���������ƵõĹ��弴ΪMg���������ڶ���ʱҪע��D��CҺ����ƽ���������彫��ѹ����ɶ�������
(4)B�з�����ӦΪ��2Al+2NaOH+2H2O=2NaAlO2+3H2�����μӷ�Ӧ����������Ϊ(a-c)g���������ΪbmL�����������ԭ������ΪM����ϵ����غ���㣻
(5)��δϴ�ӣ��ᵼ��Mg�����в������ӣ�Mg����ƫ��
(1)Mg��Al���DZȽϻ��õĽ��������߶��������ᷴӦ����ֻ��Al��NaOH��Һ��Ӧ����������Mg���ܣ����A���Լ�ΪNaOH��Һ��
(2) Mg��Al���DZȽϻ��õĽ��������ֽ���������Ӵ����ڽ������������һ��MgO��Al2O3����Ĥ��ʵ��ǰ���Ƚ���þ�Ͻ���ϡ�����н���Ƭ�̣���Ŀ�ij�ȥþ�����Ͻ���������Ĥ��
(3)ʵ��ʱ����Ҫ��������ԣ�������������C��Һ��λ�ã��ټ���NaOH��Һ��ʼ��Ӧ������Ӧ��ϲ���ȴ�����º�¼��������C��Һ��λ�ã����B��ʣ�������ˣ�ϴ�ӣ�������أ�����˳��Ϊ�ݢ٢ܢۢڣ������ܶ���ʱΪʹ��������ѹǿ��������ѹ��ȣ�����ʹD��C������Һ����ƽ��
(4) B��Al��NaOH��Һ������Ӧ�Ļ�ѧ����ʽΪ��2Al+2NaOH+2H2O=2NaAlO2+3H2�����μӷ�Ӧ����������Ϊ(a-c)g���������ΪbmL�����������ԭ������ΪM���ɵ����غ��֪��![]() �����M=
�����M=![]() ��
��
(5)��δϴ�ӹ������õIJ����cֵƫ�������(a-c)��С����������������![]() ��100%��֪��ʵ�������������������ƫС��
��100%��֪��ʵ�������������������ƫС��

 �ܿ���ȫ��100��ϵ�д�
�ܿ���ȫ��100��ϵ�д�����Ŀ��Ԫ�ص��ʼ��仯�����й㷺��;����������ڱ��е�������Ԫ�����֪ʶ�ش��������⣺
��1����ԭ������������˳��ϡ��������⣩������˵����ȷ����___��
a��ԭ�Ӱ뾶�����Ӱ뾶����С b�������Լ������ǽ�������ǿ
c�����ʵ��۵㽵�� d���������Ӧ��ˮ������Լ�����������ǿ
��2��ԭ�������������������������ͬ��Ԫ������Ϊ___�������������ļ���������___��
��3����֪��
������ | MgO | Al2O3 | MgCl2 | AlCl3 |
���� | ���ӻ����� | ���ӻ����� | ���ӻ����� | ���ۻ����� |
�۵�/�� | 2800 | 2050 | 714 | 191 |
��ҵ��þʱ�����MgCl2�������MgO��ԭ����___������ʱ�����Al2O3�������AlCl3��ԭ����___��
��4������裨�۵�1410���������õİ뵼����ϣ��ɴֹ��ƴ���������£�
Si���֣�![]() SiCl4
SiCl4![]() SiCl4������
SiCl4������![]() Si������
Si������
д��SiCl4�ĵ���ʽ��___����������SiCl4�ƴ���ķ�Ӧ�У����ÿ����1.12kg����������akJ������д���÷�Ӧ���Ȼ�ѧ����ʽ___��
��5���������岻����Ũ����������P2O5�������___��
a��NH3 b��HI c��SO2 d��CO2
��6��KClO3������ʵ������O2�������Ӵ�����400��ʱ�ֽ�ֻ���������Σ�����һ�����������Σ���һ���ε��������Ӹ�����Ϊ1��1��д���÷�Ӧ�Ļ�ѧ����ʽ��___��