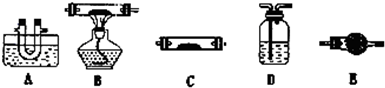
��Ŀ����
ʵ����Ʒ������ͭ���塢�в��������������������żܡ���������ҩ�ס�������ƽ
ʵ�鲽�裺
�� ȷ����һ���ɾ��������������
�� �������м���һ����������ͭ�������������أ������������������в�����ϸ���ٷŻص������У�
�� ��ʢ���������������ȣ��������ɰ�ɫ��ĩʱ��ֹͣ���ȣ�
�� ��������е������������������ȴ�����º��أ�
�� ��������е������ټ���һ��ʱ�䣬�������������ȴ�����º�������ظ���������ֱ���������γ��������������0.1gΪֹ��
�� ��������ͭ���廯ѧʽ��x��ʵ��ֵ��
�����÷������ش��������⣺
��1����ɱ�ʵ�黹��Ҫ��ʵ����Ʒ��___________ ��
��2�� ָ��ʵ�鲽����д��ڵĴ�������_______________ ��
��3������ͭ���ܷ����ڿ�������ȴ��ԭ���� _______________��
��4������ݵ�Ŀ����________________ ��
��5��������������Ϊm������������ͭ���������Ϊm1�����Ⱥ������������ˮ����ͭ������Ϊm2������CuSO4��xH2O�У�x ��______________ ��д����ʽ����
��6�����������п�����ɲ��Խ��ƫ�ߵ��� ____________������ţ���
A�������к��м��Ȳ��ӷ�������
B�������к��м����ӷ�������
C������ǰ�������в�����ˮ
D��ʵ��ǰ����δ��ȫ����
E�����������ˮ����ȫ
F������ʱ�о��彦��
��2��Ӧ�Ƚ�������ϸ���������������
��3��������ͭ�����ڿ�������ȴʱ�������տ����е�ˮ��
��4����֤������ˮ��ȫ
��5��
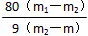
��6��B��D��F

��11�֣�������ijѧϰС����Ҷ����ijЩ���ʽ����о���ѧϰ�Ĺ��̣�
[�о�����]̽���Ҷ����ijЩ����
[��������]�Ҷ��ᣨHOOC��COOH���׳Ʋ��ᣬ����Ҫ�����������£�
| ���� | �Ҷ��� | �Ҷ��ᾧ�� |
| ����ʽ | H2C2O4 | H2C2O4��2H2O |
| ��ɫ״̬ | ��ɫ���� | ��ɫ���� |
| �ܽ�ȣ�g�� | 8.6��20�棩 | �� |
| �۵㣨�棩 | 189.5 | 101.5 |
| �ܶȣ�g��cm��3�� | 1.900 | 1.650 |
������100��ʱ��ʼ������157��ʱ��������������ʼ�ֽ⡣
����Ʋ�����ˮ��
����������ʹ����ʯ��ˮ����ǡ�
���������ڵ����¿�����Ϊ���塣
�������������ṩ����Ϣ���ش��������⣺
[�������]
������һ�����ݲ��ᾧ�����ɶ���ֽ������в���
��Ʒ�����
��1����С��ͬѧ���������ΪCO��CO2��H2O����������װ�����һ��̽��ʵ��װ�ã����ᾧ��ֽ�װ���ԣ�װ�ÿ��ظ�ʹ�ã����ӵ�����ȥ����

A��ˮ��װ��ˮ����B��װ����ͭ��C��װ��ˮ����ͭ��D��װ����ʯ��ˮ��E��װ��ʯ��
��ش��������⣺
��װ�õ�����˳��Ϊ��A��_____________________________________________��
�ڼ��������CO��ʵ��������____________________________________________________________
������װ���Ƿ���ڲ�����֮���� �����ǻ�����и���ν��___________________________________________________________________________
����������Ҷ������������
��Ʒ�����
��2����С��ͬѧΪ��֤����������������������ʵ�飬�����ܴﵽʵ��Ŀ����______������ĸ����
A�������ᾧ�����ں���̪��NaOH��Һ�У���Һ��ɫ
B���ⶨ��ͬŨ�ȵIJ����������Һ��pH
C���ⶨ�����ƣ�Na2C2O4����Һ��pH
D����������Һ����Na2CO3��Һ�У���CO2�ų�
�����������Ҷ�����л�ԭ��
��Ʒ�����
��3����С��ͬѧ���������ữ��KMnO4��Һ�е�������IJ�����Һ����������KMnO4��Һ��ɫ���Ӷ��жϲ�����н�ǿ�Ļ�ԭ�ԡ���ƽ�÷�Ӧ�����ӷ���ʽ��
___MnO4��+___H2C2O4 +___H+ ===___Mn2+ +___CO2��+___H2O
��4����������ԭ���ɶ����ⶨij���ᾧ����Ʒ������H2C2O4��2H2O������һЩ���ʣ���H2C2O4��2H2O�ĺ�����
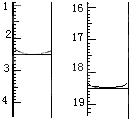
�����ǣ���ȡ����Ʒ0.12 g��������ˮ��ȫ�ܽ⣬Ȼ����0.020 mol��L��1
������KMnO4��Һ�ζ����յ㣨���ʲ����뷴Ӧ�����ζ�ǰ��ζ����е�Һ�������ͼ��ʾ����λ��mL������ò��ᾧ����Ʒ��H2C2O4��2H2O����������Ϊ_____________��
����֪���ԭ��������Mr(H2C2O4��2H2O)=126��

��11�֣�������ijѧϰС����Ҷ����ijЩ���ʽ����о���ѧϰ�Ĺ��̣�
[�о�����]̽���Ҷ����ijЩ����
[��������]�Ҷ��ᣨHOOC��COOH���׳Ʋ��ᣬ����Ҫ�����������£�
|
���� |
�Ҷ��� |
�Ҷ��ᾧ�� |
|
����ʽ |
H2C2O4 |
H2C2O4��2H2O |
|
��ɫ״̬ |
��ɫ���� |
��ɫ���� |
|
�ܽ�ȣ�g�� |
8.6��20�棩 |
�� |
|
�۵㣨�棩 |
189.5 |
101.5 |
|
�ܶȣ�g��cm��3�� |
1.900 |
1.650 |
��֪��
������100��ʱ��ʼ������157��ʱ��������������ʼ�ֽ⡣
����Ʋ�����ˮ��
����������ʹ����ʯ��ˮ����ǡ�
���������ڵ����¿�����Ϊ���塣
�������������ṩ����Ϣ���ش��������⣺
[�������]
������һ�����ݲ��ᾧ�����ɶ���ֽ������в���
��Ʒ�����
��1����С��ͬѧ���������ΪCO��CO2��H2O����������װ�����һ��̽��ʵ��װ�ã����ᾧ��ֽ�װ���ԣ�װ�ÿ��ظ�ʹ�ã����ӵ�����ȥ����

A��ˮ��װ��ˮ����B��װ����ͭ��C��װ��ˮ����ͭ��D��װ����ʯ��ˮ��E��װ��ʯ��
��ش��������⣺
�� װ�õ�����˳��Ϊ��A��_____________________________________________��
�� ���������CO��ʵ��������____________________________________________________________
�� ����װ���Ƿ���ڲ�����֮���� �����ǻ�����и���ν��___________________________________________________________________________
����������Ҷ������������
��Ʒ�����
��2����С��ͬѧΪ��֤����������������������ʵ�飬�����ܴﵽʵ��Ŀ����______������ĸ����
A�������ᾧ�����ں���̪��NaOH��Һ�У���Һ��ɫ
B���ⶨ��ͬŨ�ȵIJ����������Һ��pH
C���ⶨ�����ƣ�Na2C2O4����Һ��pH
D����������Һ����Na2CO3��Һ�У���CO2�ų�
�����������Ҷ�����л�ԭ��
��Ʒ�����
��3����С��ͬѧ���������ữ��KMnO4��Һ�е�������IJ�����Һ����������KMnO4��Һ��ɫ���Ӷ��жϲ�����н�ǿ�Ļ�ԭ�ԡ���ƽ�÷�Ӧ�����ӷ���ʽ��
___MnO4��+___H2C2O4 +___H+ ===___Mn2+ +___CO2��+___H2O
��4����������ԭ���ɶ����ⶨij���ᾧ����Ʒ������H2C2O4��2H2O������һЩ���ʣ���H2C2O4��2H2O�ĺ�����
�����ǣ���ȡ����Ʒ0.12 g��������ˮ��ȫ�ܽ⣬Ȼ����0.020 mol��L��1
������KMnO4��Һ�ζ����յ㣨���ʲ����뷴Ӧ�����ζ�ǰ��ζ����е�Һ�������ͼ��ʾ����λ��mL������ò��ᾧ����Ʒ��H2C2O4��2H2O����������Ϊ_____________��
����֪���ԭ��������Mr(H2C2O4��2H2O) =126��
=126��