��Ŀ����
�ó������ⶨ KHCO3�� Na2CO3 �����������ɣ�ÿ�γ�ȡһ����������Ʒ����ˮ�Ƴ���Һ�������еμ���ͬŨ�ȵ� Ba(OH)2 ��Һ��ÿ��ʵ�����ַ�Ӧ����Ӧǰ����Һ����仯���Բ��ƣ�ʵ���¼���±���
ʵ����� | I | II | III | IV | �� |
��Ʒ����(g) | 3.06 | 6.12 | 9.18 | 12.24 | 15.30 |
Ba(OH)2��Һ���(mL) | 300 | 300 | 300 | 300 | 300 |
���ɳ���������(g) | 5.91 | 11.82 | 17.73 | 23.64 | 23.64 |
�ش��������⣺
��1����Ʒ��KHCO3 ��Na2CO3 �����ʵ���֮��_______��
��2�������µ�III��ʵ��������Һ�е� OH-���ʵ���Ũ��Ϊ_________��
��ϰ��ϵ�д�
�����Ŀ


 2N2(g)��3H2O(g)��H<0��Ϊ��ߵ��������ת���ʿɲ�ȡ�Ĵ�ʩ�ǣ�һ�����ɣ� ��
2N2(g)��3H2O(g)��H<0��Ϊ��ߵ��������ת���ʿɲ�ȡ�Ĵ�ʩ�ǣ�һ�����ɣ� �� 2CO2(g)+N2(g)����H��0
2CO2(g)+N2(g)����H��0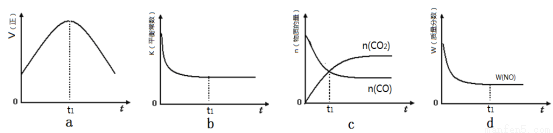

 ������11.2LN2���ɣ���״������������ClO2 g��
������11.2LN2���ɣ���״������������ClO2 g��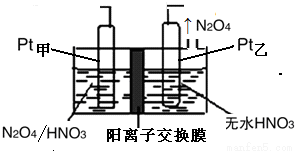
 2CO2(g)+3H2O(g) ��H1=-1366kJ��mol��1
2CO2(g)+3H2O(g) ��H1=-1366kJ��mol��1
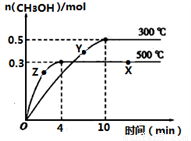
 CH3OH(g)�ﵽ��ѧƽ��״̬��
CH3OH(g)�ﵽ��ѧƽ��״̬��