ΧβΡΩΡΎ»ί
“Μ÷÷Κ§ΧΦΓΔ«βΓΔ―θ»ΐ÷÷‘ΣΥΊΒΡ”–ΜζΜ·ΚœΈοΓΘ“―÷ΣΘΚA÷–ΧΦΒΡ÷ ΝΩΖ÷ ΐΈΣ44.1%Θ§«βΒΡ÷ ΝΩΖ÷ ΐΈΣ8.82%ΘΜA÷ΜΚ§”–“Μ÷÷ΙΌΡήΆ≈Θ§«“ΟΩΗωΧΦ‘≠Ή”…œΉνΕύ÷ΜΝ§“ΜΗωΙΌΡήΆ≈ΘΚAΡή”κ““ΥαΖΔ…ζθΞΜ·Ζ¥”ΠΘ§ΒΪ≤ΜΡή‘ΎΝΫΗωœύΝΎΧΦ‘≠Ή”…œΖΔ…ζœϊ»ΞΖ¥”ΠΓΘ«κΧνΩ’ΘΚΘ®1Θ©AΒΡΖ÷Ή” Ϋ «____________Θ§ΤδΫαΙΙΦρ Ϋ «____________ΓΘ
Θ®2Θ©–¥≥ωA”κ““ΥαΖ¥”ΠΒΡΜ·―ßΖΫ≥Χ ΫΘΚ________________________ΓΘ
Θ®3Θ©–¥≥ωΥυ”–¬ζΉψœ¬Ν–3ΗωΧθΦΰΒΡAΒΡΆ§Ζ÷“λΙΙΧεΒΡΫαΙΙΦρ ΫΓΘΔΌ τ÷±Ν¥Μ·ΚœΈοΘΜΔΎ”κAΨΏ”–œύΆ§ΒΡΙΌΡήΆ≈ΘΜΔέΟΩΗωΧΦ‘≠Ή”…œΉνΕύ÷ΜΝ§“ΜΗωΙΌΡήΆ≈ΓΘ’β–©Ά§Ζ÷“λΙΙΧεΒΡΫαΙΙΦρ Ϋ «____________ΓΘ
Θ®1Θ©C5H12O4ΓΓ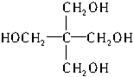
Θ®2Θ©C(CH2OH)4+4CH3COOH![]() C(CH2OCCH3)4+4H2O
C(CH2OCCH3)4+4H2O
Θ®3Θ©![]()
![]()
![]()
ΫβΈωΘΚ Ήœ»ΗυΨίΗς‘ΣΥΊΒΡ÷ ΝΩΖ÷ ΐ«σΉνΦρ ΫΘ§CΓΟHΓΟO=44.1%/12ΓΟ8.82%/1ΓΟ47.08%/16ΓΟ1=5ΓΟ12ΓΟ4Θ§”…”Ύ«β‘≠Ή”“―Ψ≠¥οΒΫ±ΞΚΆΘ§Υυ“‘ΉνΦρ ΫΨΆ «Ζ÷Ή” ΫΓΘΗυΨίAΒΡ–‘÷ ≈–ΕœΘ§A «¥ΦΘ§≤Δ«“≤ΜΡήΖΔ…ζΦ”≥…Ζ¥”ΠΘ§‘ρ”–“ΜΗωΧΦ‘≠Ή”…œΟΜ”–«β‘≠Ή”Θ§Υυ“‘AΈΣ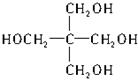 ΓΘ
ΓΘ

ΝΖœΑ≤αœΒΝ–¥πΑΗ
 »ΪΡήΝΖΩΦΨμœΒΝ–¥πΑΗ
»ΪΡήΝΖΩΦΨμœΒΝ–¥πΑΗ
œύΙΊΧβΡΩ
