��Ŀ����
�������ӷ���ʽ��д��ȷ���ǣ� ��
Ba2+ + 2OH- + NH4+ + H+ + SO42- = BaSO4��+ NH3��H2O + H2O
| A������ʯ��ˮ������С�մ���Һ��ϣ�Ca2+ + 2OH- + 2 HCO3-= CaCO3��+ CO32- + 2H2O | B����A1C13��Һ�м�������İ�ˮ��Al3++4NH3��H2O=AlO2��+4NH4++2H2O |
| C��Ba(OH)2��Һ�е���������NaHSO4��Һ��Ba2��+ 2OH��+ 2H��+ SO42��= BaSO4��+ 2H2O | |
| D����NH4HSO4ϡ��Һ����μ���Ba(OH)2ϡ��Һ���պó�����ȫ�� |
D
A������ȷ���ӷ���ʽΪ��Ca2+ + OH- + HCO3- = CaCO3��+ H2O��B������ȷ���ӷ���ʽΪ��Al3++3NH3��H2O=AL(OH)3��+3NH4+; C������ȷ���ӷ���ʽΪBa2��+ OH��+ H��+ SO42��= BaSO4��+ H2O��D��ȷ��

��ϰ��ϵ�д�
 ���б�ˢ��ϵ�д�
���б�ˢ��ϵ�д�
�����Ŀ
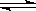 H3O+�� S2��
H3O+�� S2��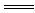 Mg2����H2O��CO2��
Mg2����H2O��CO2��
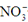 +2NO2��+2H2O
+2NO2��+2H2O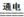 Cl2��+H2��+2OH-
Cl2��+H2��+2OH-