��Ŀ����
��1������ʵ��������Դﵽʵ��Ŀ����
a����������Һ�м���ϡ���ᣬ���ȣ���ȴ���������Cu��OH��2����ˮ�����
b�������Ը��������Һ������������ϩ
c���ñ���Na2CO3��Һ��ȥ���������е���������
d������ȼ�յķ���������ë�Ͳ�˿
��2���뽫�������ʵ�ת������뷽����������ں����ϣ�ÿһ��ֻ��дһ�ַ�������
a������b���ѽ�c������d���ѻ�e������f������g�����h������
�ٰ�úת��Ϊ��¯����ú���ͺͽ�̿��
�ڴ�ԭ���з�������͡�ú�͡����͵�
�۽�����ת��Ϊ����
�ܽ���ˮ����
�ݴӺ�ˮ�õ�����ˮMgCl2����ȡ����þ
������֬�Ʒ���
bc
bc
������ţ�����۷֣���a����������Һ�м���ϡ���ᣬ���ȣ���ȴ���������Cu��OH��2����ˮ�����
b�������Ը��������Һ������������ϩ
c���ñ���Na2CO3��Һ��ȥ���������е���������
d������ȼ�յķ���������ë�Ͳ�˿
��2���뽫�������ʵ�ת������뷽����������ں����ϣ�ÿһ��ֻ��дһ�ַ�������
a������b���ѽ�c������d���ѻ�e������f������g�����h������
�ٰ�úת��Ϊ��¯����ú���ͺͽ�̿��
e
e
���ڴ�ԭ���з�������͡�ú�͡����͵�
f
f
���۽�����ת��Ϊ����
d
d
���ܽ���ˮ����
a
a
���ݴӺ�ˮ�õ�����ˮMgCl2����ȡ����þ
g
g
��������֬�Ʒ���
h
h
����������1��a��Ӧ�ڼ��������·�Ӧ��
b����ϩ�������Ը�����ط���������ԭ��Ӧ��
c��������̼���Ʒ�Ӧ��
d����ë�Ͳ�˿����Ҫ�ɷֶ��ǵ����ʣ�
��2����ú����ɵõ���¯����ú���ͺͽ�̿�ȣ�
��ԭ�;������������͡�ú�͡����͵ȣ�
�����;��ѻ��ɵõ����ͣ�
��������ķ����õ���ˮ��
�ݹ�ҵұ����þ�õ�ⷨ��
����֬�ڼ���������ˮ��ɵõ�������
b����ϩ�������Ը�����ط���������ԭ��Ӧ��
c��������̼���Ʒ�Ӧ��
d����ë�Ͳ�˿����Ҫ�ɷֶ��ǵ����ʣ�
��2����ú����ɵõ���¯����ú���ͺͽ�̿�ȣ�
��ԭ�;������������͡�ú�͡����͵ȣ�
�����;��ѻ��ɵõ����ͣ�
��������ķ����õ���ˮ��
�ݹ�ҵұ����þ�õ�ⷨ��
����֬�ڼ���������ˮ��ɵõ�������
����⣺��1��a����������������ͭ�ķ�ӦӦ�ڼ��������·�Ӧ����a����
b����ϩ�������Ը�����ط���������ԭ��Ӧ�������ڼ��𣬹�b��ȷ��
c��������̼���Ʒ�Ӧ�����ñ���̼������Һ���ӣ���c��ȷ��
d����ë�Ͳ�˿����Ҫ�ɷֶ��ǵ����ʣ������õ�ȼ�ķ������𣬹�d����
�ʴ�Ϊ��bc��
��2������ú����ɵõ���¯����ú���ͺͽ�̿�ȣ�
��ԭ�;������������͡�ú�͡����͵ȣ�
�����;��ѻ��ɵõ����ͣ�
��������ķ����õ���ˮ��
�ݹ�ҵұ����þ�õ�ⷨ��
����֬�ڼ���������ˮ��ɵõ���������ν������Ӧ��
�ʴ�Ϊ��e��f��d��a��g��h��
b����ϩ�������Ը�����ط���������ԭ��Ӧ�������ڼ��𣬹�b��ȷ��
c��������̼���Ʒ�Ӧ�����ñ���̼������Һ���ӣ���c��ȷ��
d����ë�Ͳ�˿����Ҫ�ɷֶ��ǵ����ʣ������õ�ȼ�ķ������𣬹�d����
�ʴ�Ϊ��bc��
��2������ú����ɵõ���¯����ú���ͺͽ�̿�ȣ�
��ԭ�;������������͡�ú�͡����͵ȣ�
�����;��ѻ��ɵõ����ͣ�
��������ķ����õ���ˮ��
�ݹ�ҵұ����þ�õ�ⷨ��
����֬�ڼ���������ˮ��ɵõ���������ν������Ӧ��
�ʴ�Ϊ��e��f��d��a��g��h��
�����������ۺϿ������ʵļ��顢���𡢷��롢����Ȼ�����ѧʵ���������Ŀ�ѶȲ���ע�����������ʵ����ʣ��������ʵ���ͬ�������ʣ�

��ϰ��ϵ�д�
�����Ŀ
 ʵ���ǽ��л�ѧ�о�����Ҫ�ֶ�֮һ����ش��������⣺
ʵ���ǽ��л�ѧ�о�����Ҫ�ֶ�֮һ����ش��������⣺
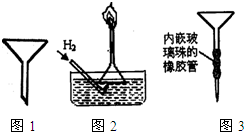 ��1������ʵ��������ʵ����ʵ��������ȷ����
��1������ʵ��������ʵ����ʵ��������ȷ����