��Ŀ����
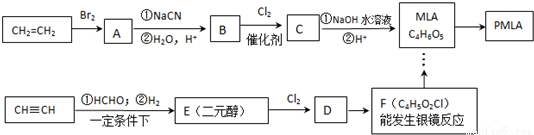
����ȫ������Ϲ����к���ƻ���ᣨMLA)�������ʽΪC4H6O5��0.1 molƻ����������NaHCO3��Һ��Ӧ�ܲ���4.48 L CO2����״������ƻ������ˮ������ʹ��ˮ��ɫ�IJ��ƻ���ᾭ�ۺ����ɾ�ƻ���ᣨPMLA����
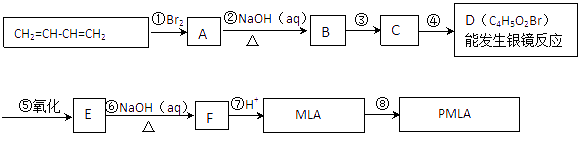
��1��д���������ʵĽṹ��ʽ��A ��D ��
��2��ָ����Ӧ���ͣ��� �� ��
��3��д��������MLA������ͬ�����ŵ�ͬ���칹��Ľṹ��ʽ�� ��
��4��д��E��Fת���Ļ�ѧ����ʽ ��
��5������ת����ϵ�в���ۺܵ͢�˳���ܷ�ߵ��� ����ܡ����ܡ���˵�����ɣ� ��
��6��PMLA�������õ����������ԣ�������Ϊ��������ߵȲ���Ӧ��������ҽҩ�������������������������ˮ��Ļ�ѧ����ʽΪ ��
��1��CH2BrCH=CHCH2Br��OHCCH2CHBrCHO
��2���ӳɣ�ȡ��
 ��3��
��3��
��4��HOOCCH2CHBrCOOH + 3NaOH �� NaOOCCH2CH(OH)COONa + NaBr + 2H2O
��5�����ܣ�����������B��̼̼˫��Ҳ��������
��6��![]() ��
��![]()
����:
���ȼ���MLA�IJ����Ͷ�Ϊ2������0.1molMLA����0.2mol������̼��˵���Ƕ�Ԫ�����������㲻���Ͷȡ���������ˮ��˵��ʣ�µ�һ����ԭ�����ǻ����ٽ�����Ʊ�ԭ��1��3-����ϩ��̼�ܽṹ����֪��ṹΪHOOCCH2CH(OH)COOH���Ա�MLA��1��3-����ϩ�Ľṹ����֪�������ʽṹ��

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�


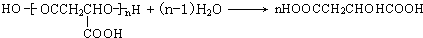



 RCH2COOH��
RCH2COOH��