��Ŀ����
��10�֣���ͼ��ʾ���ü״��������Լ�ǿ�����������Һ������ȼ�ϵ������Դ����A��Bװ��ͨ��һ��ʱ�������1.6g�״��μӷ�Ӧ�Ң۵缫���أ����蹤��ʱ��������ʧ����
��ش��������⣺
��1���ֱ�ָ��F�缫���ڵ缫������ �� ��
��2���ܵ缫�ķ�Ӧʽ�� ��E�缫�ķ�Ӧʽ�� ��
�ٵ缫�ϲ�����ʵ�������� ��
��3����A����ԭ���Һ�����Ϊ500 mL��CuSO4��K2SO4Ũ�Ⱦ�Ϊ0.1 mol/L����������A���й��ռ�����״���µ����� L��
��1�� ���� ���� (��ÿ��1�֣�
��2�� Ag ��e��=Ag+ �� CH3OH + 8OH-- 6e-="=" CO32��+ 6H2O ��
���к�ɫ�����������������ݲ��� ����ÿ��2�֣�
��3�� 3.92 ����ÿ��2�֣�
����

��ϰ��ϵ�д�
 ȫ�̽��ϵ�д�
ȫ�̽��ϵ�д�
�����Ŀ
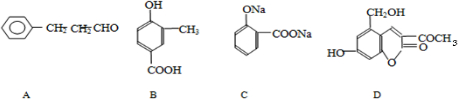
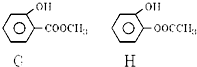


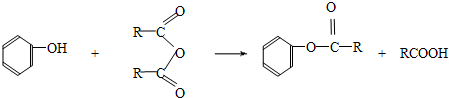
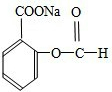



 ______��______��
______��______��
