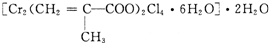��Ŀ����
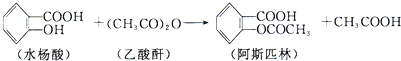
 ��2010?����һģ���������й��ס���Ч���Ͷ���ɱ�����������ǽ��չ�����ƣ�
��2010?����һģ���������й��ס���Ч���Ͷ���ɱ�����������ǽ��չ�����ƣ���1��Cl2��H2O2��ClO2����ԭ����ΪCl-����O3��1mol O3ת��Ϊ 1mol O2��1mol H2O�������ʳ��������������������ʵ�����������������Ч����ߵ���
C
C
��������ţ�A��Cl2��������B��H2O2������C��ClO2��������D��O3
��2���Ȱ���NH2Cl������Ϊɱ���������û�ѧ����ʽ��ʾ�Ȱ���ˮ�е�ɱ��������
NH2Cl+H2O=NH3+HClO
NH2Cl+H2O=NH3+HClO
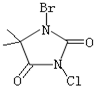
����3�����庣��C5H6BrClN2O2����һ�ֱȽϰ�ȫ�����������Ƽ��������Ʒ����ṹʽ����ͼ��ʾ��
���庣����ˮ�е�ˮ����̱Ƚϸ��ӣ���Ҫ��ˮ�����ɴ�±��ͺ����뻭������Ľṹʽ��

��4���Բ���Pt������Ϊ�缫����KOH��ҺΪ���Һ���õ�ⷨ���Ʊ�������أ�K2FeO4����
д�������ĵ缫��Ӧʽ��
Fe+8OH--6e-=FeO42-+4H2O
Fe+8OH--6e-=FeO42-+4H2O
����������1�������ʵ���ʱ����õ���Խ�࣬����Ч��Խ�ߣ�
��2��NH2Cl��ˮ��Ӧ���ɵ�HClO��NH3��HClO��ǿ�����ԣ��߱�ɱ�����ã�
��3�����庣����ˮ��ˮ����Ҫ���ɴ�±��ͺ������庣������е�±��ԭ�ӣ�Cl��Br����ˮ�е���ԭ��ȡ����Ȼ��±��ԭ����ˮ�е��ǻ���-OH�����ΪHXO��
��4�����ʱ��������������Ӧ��Fe�ڼ��������±�����ΪFeO42-��
��2��NH2Cl��ˮ��Ӧ���ɵ�HClO��NH3��HClO��ǿ�����ԣ��߱�ɱ�����ã�
��3�����庣����ˮ��ˮ����Ҫ���ɴ�±��ͺ������庣������е�±��ԭ�ӣ�Cl��Br����ˮ�е���ԭ��ȡ����Ȼ��±��ԭ����ˮ�е��ǻ���-OH�����ΪHXO��
��4�����ʱ��������������Ӧ��Fe�ڼ��������±�����ΪFeO42-��
����⣺��1�����ʵ�����Ϊ1molʱ��Cl2���2mol���ӣ�H2O2���2mol���ӣ�ClO2���5mol���ӣ�O3���2mol���ӣ�ClO2�õ��ĵ�����࣬����Ч����ߣ�
�ʴ�Ϊ��C��
��2��NH2Cl��ˮ��Ӧ���ɵ�HClO��NH3����Ӧ����ʽΪNH2Cl+H2O=NH3+HClO��HClO��ǿ�����ԣ��߱�ɱ�����ã�
�ʴ�Ϊ��NH2Cl+H2O=NH3+HClO��
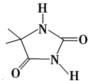
��3�����庣����ˮ��ˮ����Ҫ���ɴ�±��ͺ������庣������е�±��ԭ�ӣ�Cl��Br����ˮ�е���ԭ��ȡ����Ȼ��±��ԭ����ˮ�е��ǻ���-OH�����ΪHXO���ʺ���ĽṹʽΪ ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
��4�����ʱ��������������Ӧ��Fe�ڼ��������±�����ΪFeO42-���缫��ӦʽΪ��Fe+8OH--6e-=FeO42-+4H2O��
�ʴ�Ϊ��Fe+8OH--6e-=FeO42-+4H2O��
�ʴ�Ϊ��C��
��2��NH2Cl��ˮ��Ӧ���ɵ�HClO��NH3����Ӧ����ʽΪNH2Cl+H2O=NH3+HClO��HClO��ǿ�����ԣ��߱�ɱ�����ã�
�ʴ�Ϊ��NH2Cl+H2O=NH3+HClO��
��3�����庣����ˮ��ˮ����Ҫ���ɴ�±��ͺ������庣������е�±��ԭ�ӣ�Cl��Br����ˮ�е���ԭ��ȡ����Ȼ��±��ԭ����ˮ�е��ǻ���-OH�����ΪHXO���ʺ���ĽṹʽΪ
 ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
����4�����ʱ��������������Ӧ��Fe�ڼ��������±�����ΪFeO42-���缫��ӦʽΪ��Fe+8OH--6e-=FeO42-+4H2O��
�ʴ�Ϊ��Fe+8OH--6e-=FeO42-+4H2O��
���������⿼��������ԭ��Ӧ���л�������ʡ��绯ѧ��֪ʶ�ȣ��Ѷ��еȣ���2��Ϊ�״��㣬ע�����Ȱ���NH2Cl������Ϊɱ������ȷ������ˮ�ⷴӦ��HClO���ɣ�

��ϰ��ϵ�д�
�����Ŀ
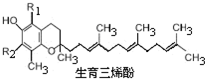 ��2010?����һģ�������£��úͦ�����������ϩ���ǹ���ά����E����Ҫ���ʣ����������в���ȷ���ǣ�������
��2010?����һģ�������£��úͦ�����������ϩ���ǹ���ά����E����Ҫ���ʣ����������в���ȷ���ǣ�������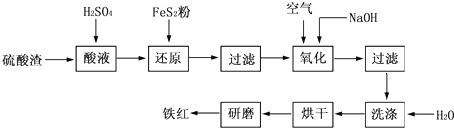
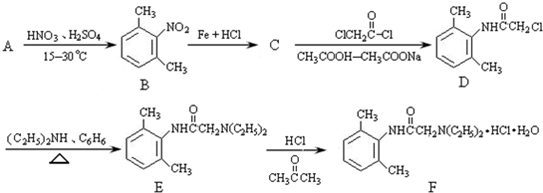
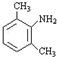
 ��������NaOH��Һ���ȳ�ַ�Ӧ�Ļ�ѧ����ʽ��
��������NaOH��Һ���ȳ�ַ�Ӧ�Ļ�ѧ����ʽ��
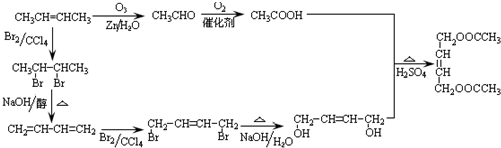
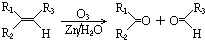
 ��2010?����һģ������Ԫ������������ͿƼ��ȷ����й㷺����;��
��2010?����һģ������Ԫ������������ͿƼ��ȷ����й㷺����;��