��Ŀ����
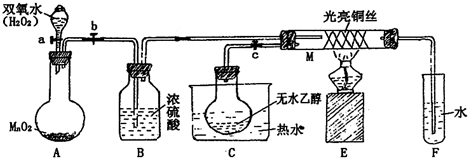
 ��2011?�Ϻ�����ͼ��ʵ������ȡHCl����NaCl+H2SO4��Ũ��
��2011?�Ϻ�����ͼ��ʵ������ȡHCl����NaCl+H2SO4��Ũ��| �� |
��1����������A��������
Բ����ƿ
Բ����ƿ
��Bƿ��Ũ����������������Ȼ�������
�����Ȼ�������
����2��Cƿ�й۲쵽��ʵ��������
�а��̲���
�а��̲���
����Ӧ�Ļ�ѧ����ʽ��NH3+HCl=NH4Cl
NH3+HCl=NH4Cl
����3��֤��HCl�����к�����Ԫ�ص�ʵ�鷽����������
������ͨ����������Һ�У��а�ɫ��������
������ͨ����������Һ�У��а�ɫ��������
����������1����ͼ��֪ʹ�õ�������B��Ũ���������ˮ�ԣ�
��2��Cƿ�гɵ�HCl�백����Ӧ�����Ȼ�泥�
��3��֤��HCl�����к�����Ԫ�أ�����ˮ���������Ӽ��ɣ�
��2��Cƿ�гɵ�HCl�백����Ӧ�����Ȼ�泥�
��3��֤��HCl�����к�����Ԫ�أ�����ˮ���������Ӽ��ɣ�
����⣺��1����ͼ��֪��������A��������Բ����ƿ��B��Ũ���������ˮ�ԣ��ɸ����Ȼ������壬�ʴ�Ϊ��Բ����ƿ�������Ȼ������壻
��2��Cƿ�гɵ�HCl�백����Ӧ�����Ȼ�泥��ɹ۲쵽�а��̲����������ķ�ӦΪNH3+HCl=NH4Cl���ʴ�Ϊ���а��̲�����NH3+HCl=NH4Cl��
��3��֤��HCl�����к�����Ԫ�أ�����ˮ���������Ӽ��ɣ�ʵ�鷽��������Ϊ������ͨ����������Һ�У��а�ɫ�����������������ӣ�����HCl�к���Ԫ�أ�
�ʴ�Ϊ��������ͨ����������Һ�У��а�ɫ����������
��2��Cƿ�гɵ�HCl�백����Ӧ�����Ȼ�泥��ɹ۲쵽�а��̲����������ķ�ӦΪNH3+HCl=NH4Cl���ʴ�Ϊ���а��̲�����NH3+HCl=NH4Cl��
��3��֤��HCl�����к�����Ԫ�أ�����ˮ���������Ӽ��ɣ�ʵ�鷽��������Ϊ������ͨ����������Һ�У��а�ɫ�����������������ӣ�����HCl�к���Ԫ�أ�
�ʴ�Ϊ��������ͨ����������Һ�У��а�ɫ����������
���������⿼���ѻӷ�����ȡ�ӷ������ʵ�飬����ʵ��װ�á����������ü�����Ŀ��飬ע������Ԫ���غ㼰�������ӵļ�����3������Ŀ�ѶȲ���

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ
 ��2011?�Ϻ�ģ�⣩��ͼ��ij��������Ķ��۷��ӣ��÷�����A��B����Ԫ�ض��ǵ������ڵ�Ԫ�أ�����������ԭ�ӵ��������Ӷ��ﵽ8�����ӵ��ȶ��ṹ������˵������ȷ���ǣ�������
��2011?�Ϻ�ģ�⣩��ͼ��ij��������Ķ��۷��ӣ��÷�����A��B����Ԫ�ض��ǵ������ڵ�Ԫ�أ�����������ԭ�ӵ��������Ӷ��ﵽ8�����ӵ��ȶ��ṹ������˵������ȷ���ǣ������� ��2011?�Ϻ���ʵ������ȡ�����������װ������ͼ��ʾ�������������������գ�
��2011?�Ϻ���ʵ������ȡ�����������װ������ͼ��ʾ�������������������գ� ��2011?�Ϻ���������ͼװ�ô������壬����ƿ�����ϣ����·�����ȷ���ǣ�������
��2011?�Ϻ���������ͼװ�ô������壬����ƿ�����ϣ����·�����ȷ���ǣ�������