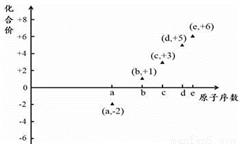
��Ŀ����
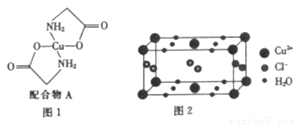
[���ʽṹ������]��֪���������İ�ˮ�ӵ�����ͭ��Һ�У���Һ���ձ������ɫ�����������Ҵ�����������ɫ�ľ���[Cu(NH3)4]SO4��H2O������ͭ��Һ�е��백�������ƣ�H2NCH2COONa���ɵõ���ͼ1��ʾ�����A��
��1��Cu2+��̬��������Ų�ʽΪ____________��
��2��N��O��S��һ�������ɴ�С��˳��Ϊ_________________��
��3��д��һ����NH3���ӻ�Ϊ�ȵ�����������ӣ�_________________��
��4�������A��̼ԭ�ӵĹ���ӻ�����Ϊ_______________��
��5��1mol ���������ƣ�H2NCH2COONa�����ЦҼ�����ĿΪ________��
��6��ij���нᾧˮ��ͭ���Ȼ���ľ����ṹ��ͼ2��ʾ���ý����Ȼ���Ļ�ѧʽ��_____����Cl-���λ�ã�4���ھ����ϣ�2���ھ����ڣ�
��ϰ��ϵ�д�
�����Ŀ