��Ŀ����
������A��B��C��D��E���ֶ�����Ԫ�ص�ijЩ����
��1��Ԫ��A���γ��л������ҪԪ�أ����з����к���sp��sp3�ӻ���ʽ����______
A��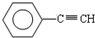 B��CH4C��CH2=CHCH3 D��CH3CH2C��CHE��CH3CH3
B��CH4C��CH2=CHCH3 D��CH3CH2C��CHE��CH3CH3
��2����AD2��Ϊ�ȵ�����ķ��ӡ����ӵĻ�ѧʽ����Ϊ______��______����д1�֣�
��3����ͬ�����£�AD2��BD2����������ˮ�е��ܽ�Ƚϴ����______��д����ʽ����������______��
| A | B | C | D | E | |
| ���ϼ� | -4 | -2 | -1 | -2 | -1 |
| �縺�� | 2.5 | 2.5 | 3.0 | 3.5 | 4.0 |
A��
 B��CH4C��CH2=CHCH3 D��CH3CH2C��CHE��CH3CH3
B��CH4C��CH2=CHCH3 D��CH3CH2C��CHE��CH3CH3��2����AD2��Ϊ�ȵ�����ķ��ӡ����ӵĻ�ѧʽ����Ϊ______��______����д1�֣�
��3����ͬ�����£�AD2��BD2����������ˮ�е��ܽ�Ƚϴ����______��д����ʽ����������______��
A��B��C��D��E���ֶ�����Ԫ�أ�����Ԫ�ػ��ϼ�֪��A���ڵڢ�A�壬B��D���ڵڢ�A�壬C��E���ڵ�V��A�壬B�ĵ縺��С��D������D��OԪ�أ�B��SԪ�أ�C�ĵ縺��С��E������C��ClԪ�أ�E��FԪ�أ���ĵ縺��С��S��A�ĵ縺�Ե���SԪ�أ�����A��CԪ�أ�
��1��A��������̼ԭ�Ӻ���3���� ��������sp2�ӻ�����A����
B�����������̼ԭ�Ӻ���4���� ��������sp3�ӻ�����B����
C����ϩ�м�̼ԭ�Ӻ���4���� ��������sp3�ӻ���̼̼˫�������̼ԭ�Ӻ���3���� ��������sp2�ӻ�����C����
D��1-��Ȳ�м����Ǽ���̼ԭ�Ӻ���4���� ��������sp3�ӻ���̼̼���������̼ԭ�Ӻ���2���� ��������sp�ӻ�����D��ȷ��
E�������м�̼ԭ�Ӻ���4���� ��������sp3�ӻ�����E����
��ѡD��
��2����CO2��Ϊ�ȵ�����ķ��ӡ������к���3��ԭ�ӣ�16���۵��ӣ�������ȵ�����ķ��ӡ����ӵĻ�ѧʽ����ΪN2O��SCN-���ʴ�Ϊ��N2O��SCN-��
��3����ͬ�����£�CO2��SO2����������ˮ�е��ܽ�Ƚϴ������������̼�ǷǼ��Է��ӣ����������Ǽ��Է��ӣ�ˮ�Ǽ��Է��ӣ����ݡ��������ܡ�ԭ��֪��SO2��H2O�е��ܽ�Ƚϴ�
�ʴ�Ϊ��SO2����ΪCO2�ǷǼ��Է��ӣ�SO2��H2O���Ǽ��Է��ӣ����ݡ��������ܡ�ԭ����SO2��H2O�е��ܽ�Ƚϴ�
��1��A��������̼ԭ�Ӻ���3���� ��������sp2�ӻ�����A����
B�����������̼ԭ�Ӻ���4���� ��������sp3�ӻ�����B����
C����ϩ�м�̼ԭ�Ӻ���4���� ��������sp3�ӻ���̼̼˫�������̼ԭ�Ӻ���3���� ��������sp2�ӻ�����C����
D��1-��Ȳ�м����Ǽ���̼ԭ�Ӻ���4���� ��������sp3�ӻ���̼̼���������̼ԭ�Ӻ���2���� ��������sp�ӻ�����D��ȷ��
E�������м�̼ԭ�Ӻ���4���� ��������sp3�ӻ�����E����
��ѡD��
��2����CO2��Ϊ�ȵ�����ķ��ӡ������к���3��ԭ�ӣ�16���۵��ӣ�������ȵ�����ķ��ӡ����ӵĻ�ѧʽ����ΪN2O��SCN-���ʴ�Ϊ��N2O��SCN-��
��3����ͬ�����£�CO2��SO2����������ˮ�е��ܽ�Ƚϴ������������̼�ǷǼ��Է��ӣ����������Ǽ��Է��ӣ�ˮ�Ǽ��Է��ӣ����ݡ��������ܡ�ԭ��֪��SO2��H2O�е��ܽ�Ƚϴ�
�ʴ�Ϊ��SO2����ΪCO2�ǷǼ��Է��ӣ�SO2��H2O���Ǽ��Է��ӣ����ݡ��������ܡ�ԭ����SO2��H2O�е��ܽ�Ƚϴ�

��ϰ��ϵ�д�
 ����ʦ��Сһ����ʦ������ҵϵ�д�
����ʦ��Сһ����ʦ������ҵϵ�д� ���100�ֵ�Ԫ�Ż�������ϵ�д�
���100�ֵ�Ԫ�Ż�������ϵ�д�
�����Ŀ

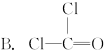
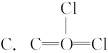 D.Cl��C��O��Cl
D.Cl��C��O��Cl
 �����Ʋ��䵥��Ϊ���غͼ״�
�����Ʋ��䵥��Ϊ���غͼ״�
 ����Ϊ4-��-2-��ϩ
����Ϊ4-��-2-��ϩ