��Ŀ����
A��B��C��D��E���ֶ�����Ԫ�أ����ǵ�ԭ��������������AԪ�ص�ԭ���ǰ뾶��С��ԭ�ӣ�BԪ�ص�����������ˮ���������⻯�ﷴӦ����һ����X��D��Aͬ���壬����Eͬ���ڣ�EԪ���������������������������
����A��B��D��E������Ԫ�أ�ÿһ����CԪ�ض��γ�Ԫ�ص�ԭ�Ӹ����Ȳ���ͬ�������ֻ������ش��������⣺
��1��д������Ԫ�ص�Ԫ�ط��ţ�B
��2���õ���ʽ��ʾD��E���γɵĻ����
 ��
��
��3��E�����̬��A��B��C����Ԫ���γɵ���ʽ��Y�Ļ�ѧʽ��
��4������ʱ��BԪ�ص�����������Ӧˮ����Z����Һ����X����Һ��pH��Ϊa������������Һ����ˮ���������H+Ũ��c1��H+����c2��H+��=
| 3 | 4 |
��1��д������Ԫ�ص�Ԫ�ط��ţ�B
N
N
��CO
O
����2���õ���ʽ��ʾD��E���γɵĻ����


��3��E�����̬��A��B��C����Ԫ���γɵ���ʽ��Y�Ļ�ѧʽ��
NH4HSO4
NH4HSO4
��������ˮ��Һ������Ũ���ɴ�С��˳����c��H+����c��SO42-����c��NH4+����c��OH-��
c��H+����c��SO42-����c��NH4+����c��OH-��
����4������ʱ��BԪ�ص�����������Ӧˮ����Z����Һ����X����Һ��pH��Ϊa������������Һ����ˮ���������H+Ũ��c1��H+����c2��H+��=
102a-14
102a-14
��������AԪ�ص�ԭ���ǰ뾶��С��ԭ�ӣ���AΪHԪ�أ�BԪ�ص�����������ˮ���������⻯�ﷴӦ����һ����X����BΪN����HNO3+NH3�TNH4NO3��D��Aͬ���壬��DΪNa����EΪ��������Ԫ�أ������������Ϊ8������������Ϊ
��8=6����EΪS������H��N��Na��S�γ�Ԫ�ص�ԭ�Ӹ����Ȳ���ͬ�����ɻ������Ԫ��CӦΪO������Ԫ���������ڱ��е�λ�ý�϶�Ӧ����������ʽ����⣮
| 3 |
| 4 |
����⣺AԪ�ص�ԭ���ǰ뾶��С��ԭ�ӣ���AΪHԪ�أ�BԪ�ص�����������ˮ���������⻯�ﷴӦ����һ����X����BΪN����HNO3+NH3�TNH4NO3��D��Aͬ���壬��DΪNa����EΪ��������Ԫ�أ������������Ϊ8������������Ϊ
��8=6����EΪS������H��N��Na��S�γ�Ԫ�ص�ԭ�Ӹ����Ȳ���ͬ�����ɻ������Ԫ��CӦΪO��
��1�������Ϸ�����֪��BΪNԪ�أ�CΪOԪ�أ��ʴ�Ϊ��N��O��
��2��DΪNaԪ�أ�EΪSԪ�أ������γɵĻ�����ΪNa2S��Ϊ���ӻ�����õ���ʽ��ʾ�γɹ���Ϊ ��
��
�ʴ�Ϊ�� ��
��
��3��E��S�������Ϊ+6����H��N��O����Ԫ���γɵ���ʽ��ΪNH4HSO4�����뷽��ʽΪNH4HSO4�TNH4++H++SO4-2��
��NH4+�ܷ���ˮ��NH4++H2O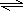 NH3?H2O+H+��������Ũ��������H+�������SO42-���ٴ���NH4+��OH-��С��ZΪHNO3��XΪNH4NO3��
NH3?H2O+H+��������Ũ��������H+�������SO42-���ٴ���NH4+��OH-��С��ZΪHNO3��XΪNH4NO3��
�ʴ�Ϊ��NH4HSO4��c��H+����c��SO42-����c��NH4+����c��OH-����
��4��BԪ�ص�����������Ӧˮ����ZΪHNO3����X����ҺΪNH4NO3��Һ��
����HNO3��Һ����pH=aʱ��ˮ�������c��H+��=c��OH-��=
=10a-14 mol?L-1��
����NH4NO3��Һ����pH=aʱ��ˮ�������H+Ũ��Ϊ10-a mol?L-1�����ߵı�ֵΪ102a-14���ʴ�Ϊ��102a-14��
| 3 |
| 4 |
��1�������Ϸ�����֪��BΪNԪ�أ�CΪOԪ�أ��ʴ�Ϊ��N��O��
��2��DΪNaԪ�أ�EΪSԪ�أ������γɵĻ�����ΪNa2S��Ϊ���ӻ�����õ���ʽ��ʾ�γɹ���Ϊ
 ��
���ʴ�Ϊ��
 ��
����3��E��S�������Ϊ+6����H��N��O����Ԫ���γɵ���ʽ��ΪNH4HSO4�����뷽��ʽΪNH4HSO4�TNH4++H++SO4-2��
��NH4+�ܷ���ˮ��NH4++H2O
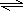 NH3?H2O+H+��������Ũ��������H+�������SO42-���ٴ���NH4+��OH-��С��ZΪHNO3��XΪNH4NO3��
NH3?H2O+H+��������Ũ��������H+�������SO42-���ٴ���NH4+��OH-��С��ZΪHNO3��XΪNH4NO3���ʴ�Ϊ��NH4HSO4��c��H+����c��SO42-����c��NH4+����c��OH-����
��4��BԪ�ص�����������Ӧˮ����ZΪHNO3����X����ҺΪNH4NO3��Һ��
����HNO3��Һ����pH=aʱ��ˮ�������c��H+��=c��OH-��=
| 10-14 |
| 10-a |
����NH4NO3��Һ����pH=aʱ��ˮ�������H+Ũ��Ϊ10-a mol?L-1�����ߵı�ֵΪ102a-14���ʴ�Ϊ��102a-14��
���������⿼��ԭ�ӽṹ��Ԫ�������ɵĹ�ϵ����Ŀ�Ѷ��еȣ���ȷ�ƶ�Ԫ�ص�����Ϊ������Ĺؼ���

��ϰ��ϵ�д�
�����Ŀ




 Ԫ�����ڱ������ڵ�һ���������ʾ�������й�A��B��C��D��E����Ԫ�ص������У���ȷ���ǣ�������
Ԫ�����ڱ������ڵ�һ���������ʾ�������й�A��B��C��D��E����Ԫ�ص������У���ȷ���ǣ�������