��Ŀ����
���б�ʾ��Ӧ��ѧ��Ӧ�����ӷ���ʽ��ȷ����
| A����KIO3����������Һ�е�KI��5I��+IO3��+3H2O =3I2+6OH�� |
B����NH4HCO3��Һ�мӹ�����NaOH��Һ�����ȣ�NH4++OH�� NH3��+H2O NH3��+H2O |
| C��������SO2ͨ���䰱ˮ�У�SO2+NH3��H2O =HSO3��+NH4+ |
| D����ϡ����ϴ���Թ��ڱڵ�������Ag+2H��+NO3��=Ag��+NO��+H2O |
C
����

��ϰ��ϵ�д�
 ��ĩ�����ϵ�д�
��ĩ�����ϵ�д�
�����Ŀ
�����������ڵ���ʵ���
| A���������� | B��ͭ | C������ | D���� |
ij��Һ����пƬ���ò���H2,�����Һ�п��Դ���������������ǣ� ��
| A��Na+��NO3-��K+��Fe2+ | B��Fe2+��Na+��SO42-��K+ |
| C��K+��I-��NO3-��H+ | D��NH4+��NO3-��Na+��HCO3- |
�����£����и���������ָ����Һ���ܴ����������
| A��pH��1����Һ�У�Fe3+��Cl����NO3-��K+ |
| B�������̪�Ժ�ɫ����Һ�У�Na+��Mg2+��AlO2-��NO3- |
| C���������۲���H2����Һ�У�Fe2+��Na+��SO42-��ClO�� |
| D��c(SO32-)��0.1 mol��L��1����Һ�У�Na+��Cl����H+��Ca2+ |
�������ӷ���ʽ��д��ȷ���ǣ� ��
| A��������þ��ϡ���ᷴӦ��H����OH��=H2O |
| B����AlCl3��Һ�м���������ˮ��Al3����3OH��=Al(OH)3�� |
| C��ͭ����ϡ���3Cu��8H����2NO3-=3Cu2����2NO����4H2O |
| D������������Һ��ͨ�����CO2��Ca2����2ClO����H2O��CO2=CaCO3����2HClO |
25��C���и����һ���ܴ����������
| A��pH=1����Һ��Na+��CO32-��Cl-��Ca2+ |
| B��ˮ�����c��H+��=1.0��10-13����Һ��K+��HCO3-��SO42-��NH4+ |
| C��c��OH-��/c��H+��=1.0��10��12����Һ��Fe2+��NO3-��K+��Cl- |
| D������Ba2+������Һ��AlO2-��Na+��NO3-��NH4+ |
����ȷ��ʾ���з�Ӧ�����ӷ���ʽ��
| A��Ũ��������м��Ӧ��2Fe+6H+=2Fe3++3H2�� |
| B������CuSO4��Һ��Ӧ��2Na+Cu2+=Cu��+2Na+ |
| C��NaHCO3��Һ��ϡH2SO4��Ӧ��CO32-+2H+=H2O+CO2�� |
| D����FeCl3��Һ�м���Mg(OH)2:3Mg(OH)2+2Fe3+=2Fe(OH)3+3Mg2+ |
ˮ��Һ���ܴ��������һ��������
| A��Na+��Al3+��Cl-��CO32- |
| B��H+��Na+��Fe2+��MnO4- |
| C��K+��Ca2+��Cl-��NO3- |
| D��K+��NH4+��OH-��SO42- |
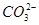 ��
��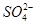 ����ȡ����100 mL��Һ��������ʵ�飺
����ȡ����100 mL��Һ��������ʵ�飺