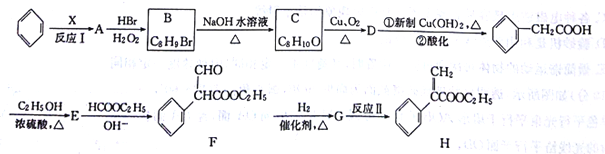
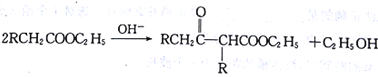��Ŀ����
����Ŀ�����Ͳ�������Fe�۾��й㷺����;��������ͨFe�۸�����������Ӧ�����Ʊ�������������ͼ��ʾ��
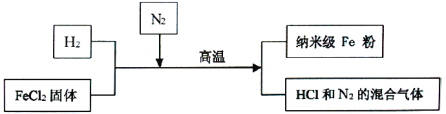
��ش��������⣺
(1)�벹����������Ʊ����յĻ�ѧ����ʽ��H2+FeCl2![]() Fe+ ________�����Ʊ�����Fe�۵Ĺ��������У�N2��������______________��
Fe+ ________�����Ʊ�����Fe�۵Ĺ��������У�N2��������______________��
(2)�о���Ա��������Ƶõ�����Fe����Ʒ�л���������FeCl2���ʡ�
��Ϊ��ȥ��Ʒ�е����ʣ������������£��ȼ�______�ܽ⣬Ȼ����______��ϴ�ӡ���ɡ�
��ȡ��Ʒ20g���������������ᣬ��������0.7g����������Ʒ�е���Fe����������Ϊ_________��
���𰸡�2HCl������ˮ����98%
��������
(1)��Ӧ��ķ����к���2����ԭ�ӡ�һ����ԭ�ӡ�2����ԭ�ӣ����������Ѿ�����һ����ԭ�ӣ���ȱ��2����ԭ�Ӻ�2����ԭ�ӣ����Բ����������2HCl��������ѧ�����ȶ����ڷ�Ӧ���𱣻��������ã��ʴ�Ϊ��2HCl����������
(2)����������ˮ���Ȼ���������ˮ�����Լ�ˮ�ܽ⣬�ٽ��й��ˡ�ϴ�Ӹ��T�ɣ��ʴ�Ϊ��ˮ�����ˣ�
��������0.7g������Ҫ�μӷ�Ӧ����������ΪX����
Fe+2HCl�TFeCl2+H2��
56 2
X 0.7g
���ݣ�![]() ��
��![]() �����X=19.6g����Ʒ�е���Fe����������Ϊ
�����X=19.6g����Ʒ�е���Fe����������Ϊ![]() ��100%=98%���ʴ�Ϊ��98%��
��100%=98%���ʴ�Ϊ��98%��