��Ŀ����
����ͪ����ɫ��dz��ɫ��Һ�壬��ǿ�ҵĴ̼��Գ�ζ���ܶȣ����ˮ=1����0.95���۵㣺-45�棬�е㣺155�棬�ܽ�ȣ�100mL H2O����2.4g��31�棩��


����Ӧ��
�ش��������⣺
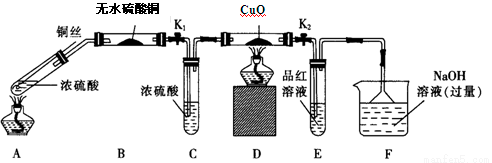
��1������A�������� ��
��2���ڷ�Ӧ��ʼ��5min��Ϊ�˽���ϵ�¶�ά����30��35�棬�����ñ�ˮԡ��ȴ�⣬��ȡ�Ĵ�ʩ���� �� ��
��3�����뱥��NaHSO3��Һʱ��������Ҫ��Ӧ�� �������ӷ���ʽ��ʾ����ȷ������ı���NaHSO3��Һ�Ѿ�������ʵ������� ��
��4��Ϊ�����Һ��pH�������ԣ����Լ�����Լ��� ��
A��ϡ���� | B����ˮ̼���� | C��Ũ���� | D���������ƹ��� |
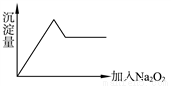
��5������pH������뾫��ʳ�Σ���Ŀ���� ����ȡ�л��㡱ʱʹ�õ���Ҫ������ ��������ͼ��Ӧ����ĸ����
��6��ʵ���������Ż���ʵ��ɰ�������Ҫ��С���Ա�����б�Ҫ��NaClO��Һ�����Ũ�Ƚ���̽��������������һϵ�в�ͬŨ�ȵ�NaClO��Һ�����õζ����궨�����巽���ǣ�����Һ��ȡ10.00mL NaClO��Һ��500mL����ƿ�ж��ݣ�ȡ25.00mL����ƿ�У���������ϡ�����KI���壨����������0.1000mol/L Na2S2O3����Һ�ζ����յ㣨�õ�����Һ��ָʾ����������ƽ��ʵ��ƽ������Na2S2O3����Һ18.00mL����֪I2+2Na2S2O3==2NaI+Na2S4O6 �����NaClO��Һ��Ũ���� ��
 ֥�鿪���γ�������ϵ�д�
֥�鿪���γ�������ϵ�д�����ʵ������������Ӧ�Ľ��۴������
ѡ�� | ʵ����������� | ���� |
A | ����ҺX�еμ�BaCl2��Һ���а�ɫ�������� | ��ҺX�п��ܺ���SO42- |
B | ���ʵ���֮��Ϊ2:3��ϡ�����ϡ����
| ��Ӧ��������ƿ����Һ��������CuSO4������ƿ���ռ�����������NO |
C | ��1mLŨ�Ⱦ�Ϊ0.05mol��L-1NaCl��NaI�Ļ����Һ�еμ�2��0.01mol��L-1AgNO3��Һ���������ǻ�ɫ | Ksp(AgCl)<Ksp(AgI) |
D | �����£���pH��ֽ���0.1mol��L-1 NaHSO3��Һ��pHԼΪ5 | HSO3-�ĵ���̶ȴ�����ˮ��̶� |
A. A B. B C. C D. D



 ��Na2S2O3��Һ�ζ����ɵ�I2��3��ʵ��ƽ�����ĸ�Na2S2O3��Һ25��00 mL������Ӧ����ͭ������Ϊ6��4 g��������ͭ�IJ���Ϊ ��
��Na2S2O3��Һ�ζ����ɵ�I2��3��ʵ��ƽ�����ĸ�Na2S2O3��Һ25��00 mL������Ӧ����ͭ������Ϊ6��4 g��������ͭ�IJ���Ϊ �� )
)