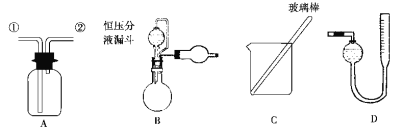
��Ŀ����
����Ŀ��
��1��ijͬѧ��������ƽ����п��24.4g(1g����������)������п���������̣�����������̣�����ƽƽ��ʱ������ȡ��п����ʵ������Ӧ��_______________g
��2������һ�����ĸ���������ԭ�Ӹ�������С�����˳�����е���
��0.5mol���� ����״����22.4L������̼ ��4��ʱ9gˮ ��0.2mol����
A���٢ܢۢ� | B���ܢۢ٢� | C���ڢۢܢ� | D���٢ܢڢ� |
��3��ʵ���Ҽ�����Һ������SO42-�ķ����ǣ�ȡ������Һ���Թ��У��ȼ�_____________�����_____________��������ְ�ɫ��������֤����SO42-
��4��0.6mol��������0.4mol����O3����ԭ�Ӹ���֮��Ϊ______________
���𰸡���1��23.6 ��2�� B ��3�� ϡ���� BaCl2��Һ ��4��1:1
�������������������1�������̵�����=���̵�����+�����������֪����������=ҩƷ����+���������������ҩƷ����=��������-������������ʵ�ʳƵ�þ�۵�����=24g-0.4g=23.6g��
��2����0.5mol��������ԭ�����ʵ���=0.5mol��4=2mol������״����22.4L������̼�����ʵ���Ϊ1mol��Ϊ��ԭ�ӷ��ӣ�����ԭ��Ϊ3mol����4��ʱ9mLˮ������Ϊ9g�������ʵ���=![]() =0.5mol������ԭ�����ʵ���=0.5mol��3=1.5mol����0.2mol�����Ậ��ԭ�����ʵ���=0.2mol��7=1.4mol���ʺ���ԭ����Ŀ������������������ѡB��
=0.5mol������ԭ�����ʵ���=0.5mol��3=1.5mol����0.2mol�����Ậ��ԭ�����ʵ���=0.2mol��7=1.4mol���ʺ���ԭ����Ŀ������������������ѡB��
��3��ȡ������Һ���Թ��У��ȼ�ϡ�����ữ�����BaCl2��Һ��������ְ�ɫ��������֤����SO42-��
��4��ÿ�����������к���2����ԭ�ӣ�ÿ�����������к���3����ԭ�ӣ�����ԭ�Ӹ�����Ϊ(3��2)��(2��3)=1��1��