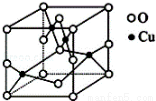
��Ŀ����
��B��C��D��E��F ��ԭ��������������Ķ���������Ԫ�أ�B �Ƕ������н�������ǿ�� Ԫ�أ�C ��ͬ���������Ӱ뾶��С��Ԫ�أ�D Ԫ�ص�����������Ӧˮ������ B Ԫ�ص���� ���������Ӧˮ���ﷴӦ�IJ��� M ���Ʊ�ľ�ķ������ԭ�ϣ�E ���������������ڲ������ ֮��Ϊ 3�s5����ش�
(1)D ��ԭ�ӽṹʾ��ͼΪ___________________��
(2)д����ҵ�� F ���ʵĻ�ѧ����ʽ��___________________��
(3)�õ���ʽ��ʾԪ�� B ��Ԫ�� F �γɵĻ����___________________��
(4)B������������ Ӧ�IJ����� C �ĵ���ͬʱ����ˮ�У�����������ɫ���壬�йصĻ�ѧ����ʽΪ____________________��___________________��
(5)д�� M �����ᷴӦ�����ӷ���ʽ��___________________��
(6)��ҵ�Ͻ������ F ����ͨ�����ڵ� E �����п��Ƶû����� E2F2�������ʿ���ˮ��Ӧ���� һ����ʹƷ����Һ��ɫ�����壬0.2mol �����ʲμӷ�Ӧʱת�� 0.3mol ���ӣ�����ֻ��һ��Ԫ�ػ��ϼ۷����ı䣬�÷�Ӧ�Ļ�ѧ����ʽΪ___________________��
��ϰ��ϵ�д�
 �ľ�ͼ���ʱ�ȷ�ϵ�д�
�ľ�ͼ���ʱ�ȷ�ϵ�д�
�����Ŀ


 2NH3 ( g ) ��H = -92.0 kJ/mol�������¶��µ�1 mol N2 ��3 mol H2 ����һ�ܱ������У��ڴ�������ʱ���з�Ӧ����÷�Ӧ�ų�������Ϊ(����������ʧ)
2NH3 ( g ) ��H = -92.0 kJ/mol�������¶��µ�1 mol N2 ��3 mol H2 ����һ�ܱ������У��ڴ�������ʱ���з�Ӧ����÷�Ӧ�ų�������Ϊ(����������ʧ)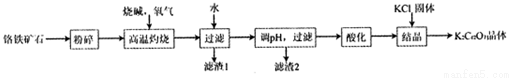
 Cr2O72-(aq)+H2O����25��ʱ��ȡNa2CrO4��Һ�����ữʵ�飬��ò���ʵ����������:
Cr2O72-(aq)+H2O����25��ʱ��ȡNa2CrO4��Һ�����ữʵ�飬��ò���ʵ����������: