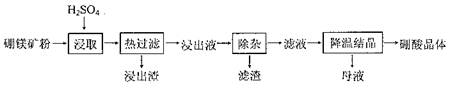
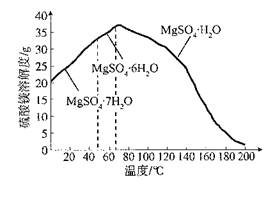
��Ŀ����
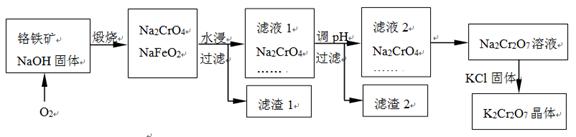
���������Ҫ�ɷֿɱ�ʾΪFeO��Cr2O3��������SiO2��Al2O3�����ʣ��Ը�����Ϊԭ���Ʊ��ظ���أ�K2Cr2O7���Ĺ�������ͼ��ʾ��
��֪���� NaFeO2��ˮǿ��ˮ�⡣ �� Cr2O72��+ H2O 2CrO42�� + 2H+
2CrO42�� + 2H+
 K2Cr2O7��CrԪ�صĻ��ϼ��� ��
K2Cr2O7��CrԪ�صĻ��ϼ��� ��
��������1�к��ɫ���ʷ�Ӧ�Ļ�ѧ����ʽ�� ��
��Һ1�ijɷֳ�Na2CrO4��Na2SiO3�⣬�����У��ѧʽ�� ��
��������2�����Ƶ��������������һ�������ᆳ���ұ���ɻ�ý������õ�ⷴӦ�Ļ�ѧ����ʽ�� ��
����Һ2ת��ΪNa2Cr2O7��ҺӦ��ȡ�Ĵ�ʩ�� ��
��Na2Cr2O7��Һ�м���KCl���壬���K2Cr2O7����IJ��������ǣ�����Ũ���� �����ˡ�ϴ�ӡ����
���ո���������Na2CrO4��NaFeO2��Ӧ�Ļ�ѧ����ʽ�� ��
��֪���� NaFeO2��ˮǿ��ˮ�⡣ �� Cr2O72��+ H2O
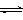 2CrO42�� + 2H+
2CrO42�� + 2H+ K2Cr2O7��CrԪ�صĻ��ϼ��� ��
K2Cr2O7��CrԪ�صĻ��ϼ��� ����������1�к��ɫ���ʷ�Ӧ�Ļ�ѧ����ʽ�� ��
��Һ1�ijɷֳ�Na2CrO4��Na2SiO3�⣬�����У��ѧʽ�� ��
��������2�����Ƶ��������������һ�������ᆳ���ұ���ɻ�ý������õ�ⷴӦ�Ļ�ѧ����ʽ�� ��
����Һ2ת��ΪNa2Cr2O7��ҺӦ��ȡ�Ĵ�ʩ�� ��
��Na2Cr2O7��Һ�м���KCl���壬���K2Cr2O7����IJ��������ǣ�����Ũ���� �����ˡ�ϴ�ӡ����
���ո���������Na2CrO4��NaFeO2��Ӧ�Ļ�ѧ����ʽ�� ��
+6 ��1�֣� ��NaFeO2+2H2O��Fe(OH)3��+NaOH�� NaOH��NaAlO2
2Al2O3(����) 4Al + 3O2�� �������ữ����ȴ�ᾧ ��1�֣�
4Al + 3O2�� �������ữ����ȴ�ᾧ ��1�֣�
4FeO��Cr2O3 + 7O2 + 20NaOH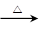 8Na2CrO4 + 4NaFeO2+ 10H2O
8Na2CrO4 + 4NaFeO2+ 10H2O
2Al2O3(����)
 4Al + 3O2�� �������ữ����ȴ�ᾧ ��1�֣�
4Al + 3O2�� �������ữ����ȴ�ᾧ ��1�֣�4FeO��Cr2O3 + 7O2 + 20NaOH
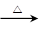 8Na2CrO4 + 4NaFeO2+ 10H2O
8Na2CrO4 + 4NaFeO2+ 10H2O�����������1�����ݻ����ﻯ�ϼ۵Ĵ�����Ϊ0��֪��K2Cr2O7��CrԪ�صĻ��ϼ��ǣ���2��7��1��2����2����6�ۡ�
��2������NaFeO2��ˮǿ��ˮ�⣬����������1�к��ɫ���ʷ�Ӧ�Ļ�ѧ����ʽ��NaFeO2+2H2O��Fe(OH)3��+NaOH�����ڿ�ʯ�л������������������������ǹ����ģ�����Һ1�ijɷֳ�Na2CrO4��Na2SiO3�⣬������NaOH��NaAlO2��
��3��������Һ�к���ƫ�����ƣ�������Ʊ���������������ڵ������������Ʊ�����������Ӧ�Ļ�ѧ����ʽ��2Al2O3(����)
 4Al + 3O2��.
4Al + 3O2��.��4�����ݷ���ʽCr2O72��+ H2O
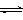 2CrO42�� + 2H+��֪������Һ2ת��ΪNa2Cr2O7��ҺӦ��ȡ�Ĵ�ʩ�Ǽ������ữ�����K2Cr2O7����IJ��������ǣ�����Ũ������ȴ�ᾧ�����ˡ�ϴ�ӡ����
2CrO42�� + 2H+��֪������Һ2ת��ΪNa2Cr2O7��ҺӦ��ȡ�Ĵ�ʩ�Ǽ������ữ�����K2Cr2O7����IJ��������ǣ�����Ũ������ȴ�ᾧ�����ˡ�ϴ�ӡ������5������ԭ���غ��֪�����ո���������Na2CrO4��NaFeO2��Ӧ�Ļ�ѧ����ʽ��4FeO��Cr2O3 + 7O2 + 20NaOH
 8Na2CrO4 + 4NaFeO2+ 10H2O��
8Na2CrO4 + 4NaFeO2+ 10H2O�������������Ǹ߿��еij������ͣ��Ѷȴ��ۺ���ǿ����ѧ����Ҫ��ߡ�������ע�ضԻ���֪ʶ���̺�ѵ����ͬʱ�����ض�ѧ�������������ͽ��ⷽ����ָ����ѵ��������������ѧ���淶�Ͻ���ʵ����������Լ��������������������ۺ���ǿ�����ۺ�ʵ������ϵ���ܣ��еĻ��ṩһЩ�µ���Ϣ�����Ҫ��ѧ���������桢ϸ�µ����⣬��ϵ��ѧ����֪ʶ�ͼ��ܣ�����֪ʶ����ȡ�Ǩ�ơ����飬ȫ��ϸ�µ�˼�����ܵó���ȷ�Ľ��ۡ�

��ϰ��ϵ�д�
�����Ŀ