��Ŀ����
�ȡ��塢��ͬ�������ڱ��еĢ�A�壬���ϸ����ͣ����Ǻ�����Ƶøߵ��ἰ���Σ����ò�����+7����Ļ����1968�꣬��ѧ���÷��ֲ��õ�XeF2��KBrO3�����Ƶô�����KBrO4���壮
��1���ߵ����Σ�IO65-���������������½�Mn2+����ΪMnO4-����������ԭ��I03-�����䷴Ӧƽ�ȶ�Ѹ�٣�д�����ӷ�Ӧ����ʽ��______��
��2����25�棬��֪pH=2�ĸߵ�����Һ��pH=12��NaOH��Һ�������ϣ����û��Һ�����ԣ�0.01mol-L-1�ĵ�����������Һ��pH=12��NaOH��Һ�������ϣ����û��Һ�����ԣ���ߵ�����______����������______���ǿ�ᡱ�����ᡱ����
��3��д��XeF2�ͺ�KOH��KBrO3��ˮ��Һ��Ӧ�õ�KBrO4�����ӷ���ʽ______��
��1���ߵ����Σ�IO65-���������������½�Mn2+����ΪMnO4-����������ԭ��I03-�����䷴Ӧƽ�ȶ�Ѹ�٣�д�����ӷ�Ӧ����ʽ��______��
��2����25�棬��֪pH=2�ĸߵ�����Һ��pH=12��NaOH��Һ�������ϣ����û��Һ�����ԣ�0.01mol-L-1�ĵ�����������Һ��pH=12��NaOH��Һ�������ϣ����û��Һ�����ԣ���ߵ�����______����������______���ǿ�ᡱ�����ᡱ����
��3��д��XeF2�ͺ�KOH��KBrO3��ˮ��Һ��Ӧ�õ�KBrO4�����ӷ���ʽ______��
��1���ߵ�������ӵõ�������������������ʧ��������ԭ���������������¸���������Ӻ������ӷ���������ԭ��Ӧ���ɵ�������Ӻ���������ӡ�ˮ���������ӷ���ʽΪ5IO65-+2Mn2++14H+=5IO3-+2MnO4-+7H2O��
�ʴ�Ϊ��5IO65-+2Mn2++14H+=5IO3-+2MnO4-+7H2O��
��2��pH=2�ĸߵ�����Һ��pH=12��NaOH��Һ��������Ũ�Ⱥ����������ӵ�Ũ����ȣ�����������ǿ���������ʱ������Һ�����ԣ�˵�������Ӻ�����������ǡ�÷�Ӧ���ߵ�����ǿ�ᣬ��ʵ���ϻ����Һ�����ԣ���˵���ߵ����ֵ�������������ӣ����Ըߵ��������ᣮ
pH=12��NaOH��Һ���������ӵ�Ũ��Ϊ0.01mol?L-1����0.01mol?L-1�ĵ�����������Һ��pH=12��NaOH��Һ��������ʱ����Һ�����ԣ�˵�������������������ӵ����ʵ��������������ӵ����ʵ�����ȣ���Һ�������ȣ�����������ǿ����Ը�����ȫ����������ǿ����ʣ�������ǿ�ᣮ
�ʴ�Ϊ���ߵ����������������ǿ�ᣮ
��3������KBrO3��KBrO4�е���Ļ��ϼ۷ֱ���+5�ۡ�+7�ۣ�������KBrO3����KBrO4��KBrO3ʧ��������ԭ��������XeF2�õ�������������FԪ�صķǽ�������ǿ������ֻ���Ի���̬���ڣ�ֻ����XeԪ�صõ������ɵ���Xe��ͬʱ��ˮ���ɣ��������ӷ���ʽΪXeF2+2OH-+BrO3-=BrO4-+Xe+2F-+H2O��
�ʴ�Ϊ��XeF2+2OH-+BrO3-=BrO4-+Xe+2F-+H2O��
�ʴ�Ϊ��5IO65-+2Mn2++14H+=5IO3-+2MnO4-+7H2O��
��2��pH=2�ĸߵ�����Һ��pH=12��NaOH��Һ��������Ũ�Ⱥ����������ӵ�Ũ����ȣ�����������ǿ���������ʱ������Һ�����ԣ�˵�������Ӻ�����������ǡ�÷�Ӧ���ߵ�����ǿ�ᣬ��ʵ���ϻ����Һ�����ԣ���˵���ߵ����ֵ�������������ӣ����Ըߵ��������ᣮ
pH=12��NaOH��Һ���������ӵ�Ũ��Ϊ0.01mol?L-1����0.01mol?L-1�ĵ�����������Һ��pH=12��NaOH��Һ��������ʱ����Һ�����ԣ�˵�������������������ӵ����ʵ��������������ӵ����ʵ�����ȣ���Һ�������ȣ�����������ǿ����Ը�����ȫ����������ǿ����ʣ�������ǿ�ᣮ
�ʴ�Ϊ���ߵ����������������ǿ�ᣮ
��3������KBrO3��KBrO4�е���Ļ��ϼ۷ֱ���+5�ۡ�+7�ۣ�������KBrO3����KBrO4��KBrO3ʧ��������ԭ��������XeF2�õ�������������FԪ�صķǽ�������ǿ������ֻ���Ի���̬���ڣ�ֻ����XeԪ�صõ������ɵ���Xe��ͬʱ��ˮ���ɣ��������ӷ���ʽΪXeF2+2OH-+BrO3-=BrO4-+Xe+2F-+H2O��
�ʴ�Ϊ��XeF2+2OH-+BrO3-=BrO4-+Xe+2F-+H2O��

��ϰ��ϵ�д�
�����Ŀ
 ��2009?�Ϻ�����������Դ�ı��⣬�̲��ŷḻ�Ļ�ѧԪ�أ����ȡ��塢��ȣ�
��2009?�Ϻ�����������Դ�ı��⣬�̲��ŷḻ�Ļ�ѧԪ�أ����ȡ��塢��ȣ�
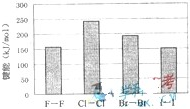
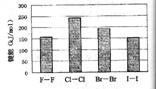 (4)±�ص��ʵļ��ܴ�С����ͼ����ͼ�ƶϣ�
(4)±�ص��ʵļ��ܴ�С����ͼ����ͼ�ƶϣ� ����ʽΪ ����Ӧ���б��ƻ��Ļ�ѧ����
����ʽΪ ����Ӧ���б��ƻ��Ļ�ѧ���� �� ��(����ԡ��Ǽ��ԡ�)��
�� ��(����ԡ��Ǽ��ԡ�)�� λ�����ڱ��ĵ�
λ�����ڱ��ĵ�  3)±�ص��ʼ���
3)±�ص��ʼ��� ���������������϶������ŵݱ���ɡ������й�˵����ȷ���� ��
���������������϶������ŵݱ���ɡ������й�˵����ȷ���� �� ��Ļ�ԭ��HF��HCl��HBr��HI��˳�����μ���
��Ļ�ԭ��HF��HCl��HBr��HI��˳�����μ���